Chapter 20: Q26P (page 1038)
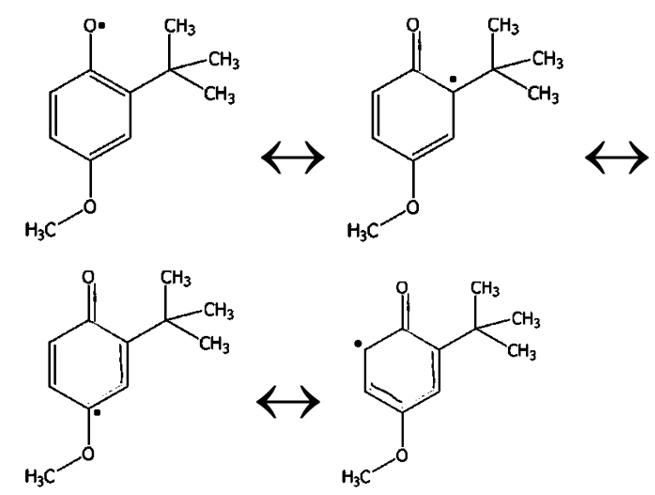
Draw resonance forms to show how the BHA radical is stabilized by delocalization of the radical electron over other atoms in the molecule.
Short Answer
Learning Materials
Features
Discover
Chapter 20: Q26P (page 1038)
Draw resonance forms to show how the BHA radical is stabilized by delocalization of the radical electron over other atoms in the molecule.

All the tools & learning materials you need for study success - in one app.
Get started for free
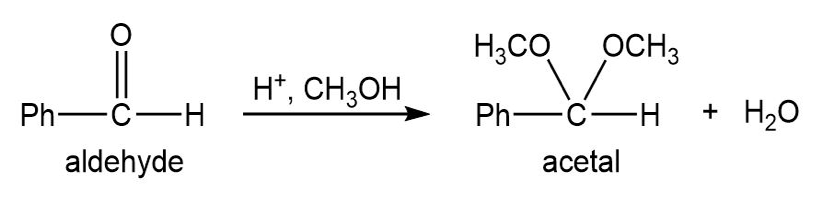
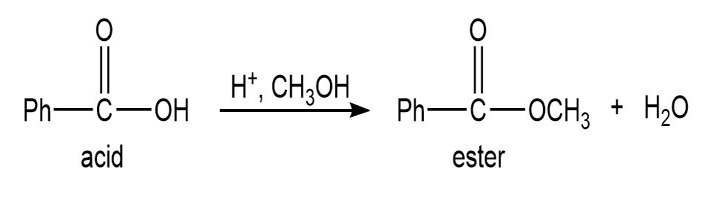
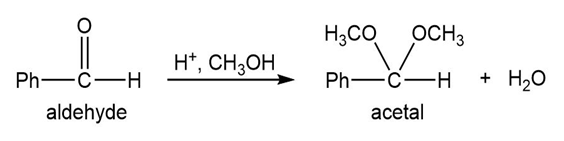
Most of the Fischer esterification mechanism is identical with the mechanism of acetal formation. The difference is in the final step, where a resonance-stabilized carbocation loses a proton to give the ester. Write mechanisms for the following reactions, with the comparable steps directly above and below each other. Explain why the final step of the esterification (proton loss) cannot occur in acetal formation, and show what happens instead.
Question: Arrange each group of compounds in order of increasing basicity.
Q.13Most of the Fischer esterification mechanism is identical with the mechanism of acetal formation. The difference is in the final step, where a resonance-stabilized carbocation loses a proton to give the ester. Write mechanisms for the following reactions, with the comparable steps directly above and below each other. Explain why the final step of the esterification (proton loss) cannot occur in acetal formation, and show what happens instead.
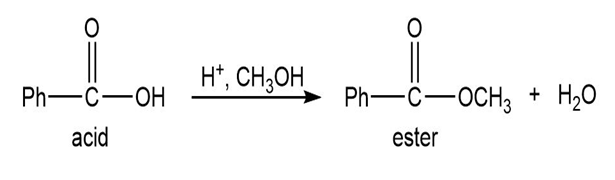
Show how you would synthesize the following compounds from the appropriate carboxylic acids or acid derivatives.
a)
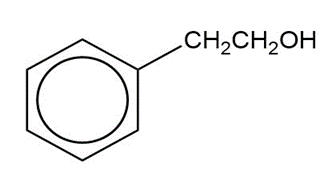
b)
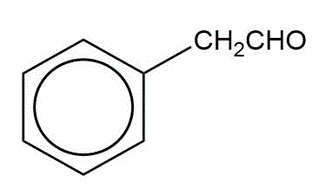
c)
Show how you would convert glycine to following derivatives. Show the structure of the product in each case.
(a) Glycine isopropyl ester
(b) N-benzoylglycine
(c) N-benzyloxycarbonylglycine
(d) Tert-butyloxycarbonylglycine
What do you think about this solution?
We value your feedback to improve our textbook solutions.