Tributylin hydride (Bu3SnH)is used synthetically to reduce alkyl halides, replacing a halogen atom with hydrogen. Free-radical initiators promote this reaction, and free-radical inhibitors are known to slow or stop it. You job is to develop a mechanism, using the following reaction as an example.
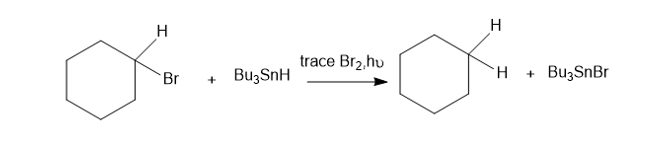
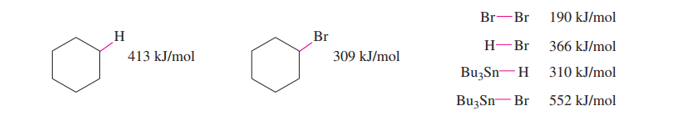
The following bond-dissociation enthalpies may be helpful:
(a) Propose initiation and propagation steps to account for this reaction.
(b) Calculate values of Δ�� for your proposed steps to show that they are energetically feasible. (Hint: A trace of Br2 and light suggests it’s there only as an initiator, to create Brradicals. Then decide which atom can be extracted most favorably from the starting materials by the Brradical. That should complete the initiation. Finally, decide what energetically favored propagation steps will accomplish the reaction).













