Chapter 21: Q60P (page 1142)
Explain this curious result. What does this reaction tell you about the relative reactivity of esters and ketones?
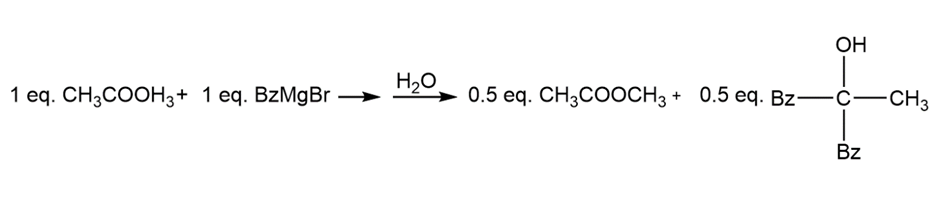
Short Answer
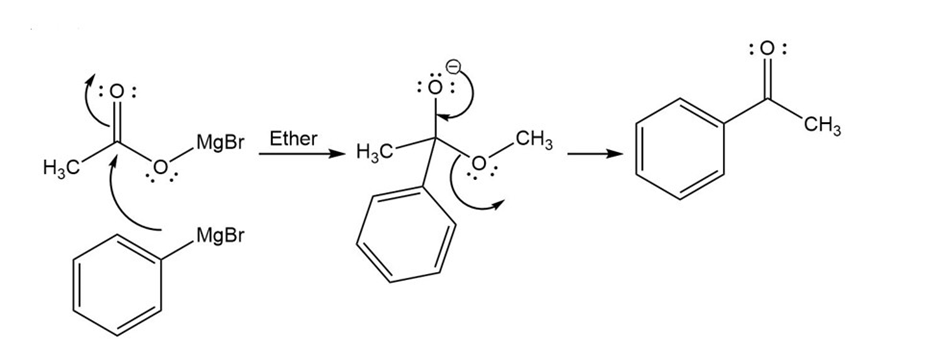
Mechanism for reaction
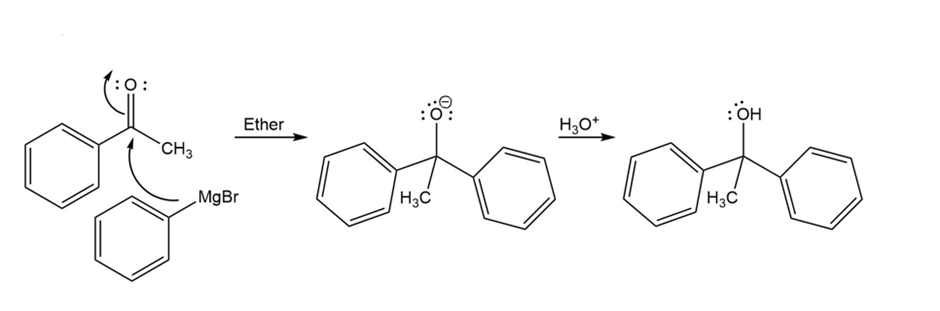
Formation of the desired product
Learning Materials
Features
Discover
Chapter 21: Q60P (page 1142)
Explain this curious result. What does this reaction tell you about the relative reactivity of esters and ketones?


Mechanism for reaction

Formation of the desired product
All the tools & learning materials you need for study success - in one app.
Get started for free
Propose a mechanism for the formation of 9-hydroxynonanoic acid lactone, as show in the preceding figure.
Show how you would use anhydrides to synthesize the following compounds. In each
case, explain why an anhydride might be preferable to an acid chloride.
(d) succinic acid monomethyl ester
Which of the following proposed reactions would take place quickly under mild conditions?
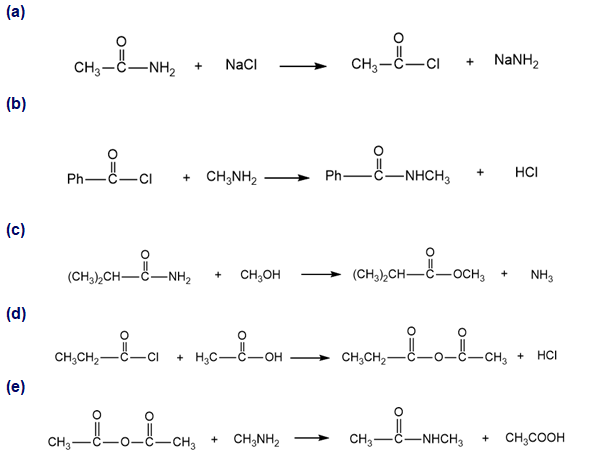
Show how you would use anhydrides to synthesize the following compounds. In each
case, explain why an anhydride might be preferable to an acid chloride.
(b) n-octyl acetate
Show how you would synthesize each compound, starting with an ester containing nomore than eight carbon atoms. Any other necessary reagents may be used.
g.
What do you think about this solution?
We value your feedback to improve our textbook solutions.