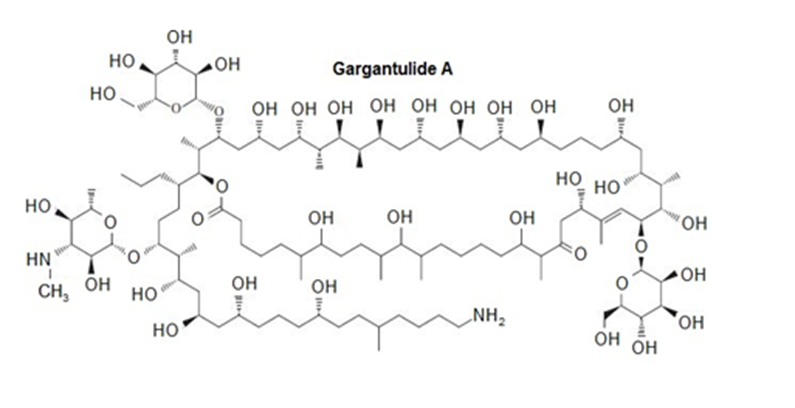
Macrolide antibiotics, including erythromycin and azithromycin (), contain a large ring lactone. One of the largest ever reported is Gargantulide A, the structure of which was determined by the research group of Prof. William Gerwick of the Scripps Institution of Oceanography (Organic Letters, 2015, 17, 1377–1380). Isolated from a Streptomyces bacterium, it kills pathogenic bacteria like MRSA and Clostridium difficile, but it proved too toxic to the test animals to continue further testing.
(a) Identify the lactone that makes this a macrolide structure.
(b) How many rings does this structure contain? How many atoms are in the largest ring?
(c) Many complex NMR experiments were used to determine the stereochemistry of most, but not all, of the chiral centers. How many chiral centers does the structure contain?