Chapter 21: Q.33P (page 1123)
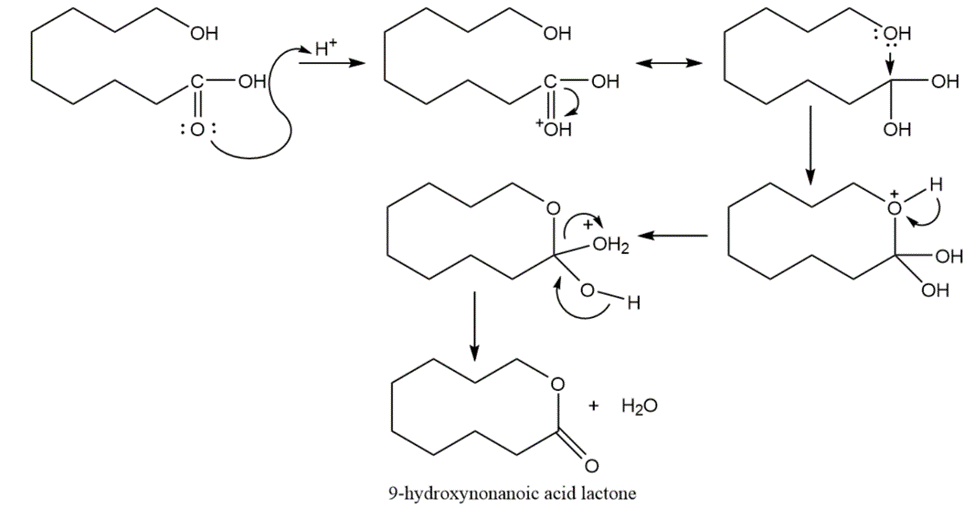
Propose a mechanism for the formation of 9-hydroxynonanoic acid lactone, as show in the preceding figure.
Short Answer
Answer
A mechanism for the formation of 9-hydroxynonanoic acid lactone is shown below.
Learning Materials
Features
Discover
Chapter 21: Q.33P (page 1123)
Propose a mechanism for the formation of 9-hydroxynonanoic acid lactone, as show in the preceding figure.
Answer
A mechanism for the formation of 9-hydroxynonanoic acid lactone is shown below.

All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would synthesize each compound, starting with an ester containing no more than eight carbon atoms. Any other necessary reagents may be used.
d.Ph2CHOH
Show how you would convert the following starting materials to the indicated nitriles:
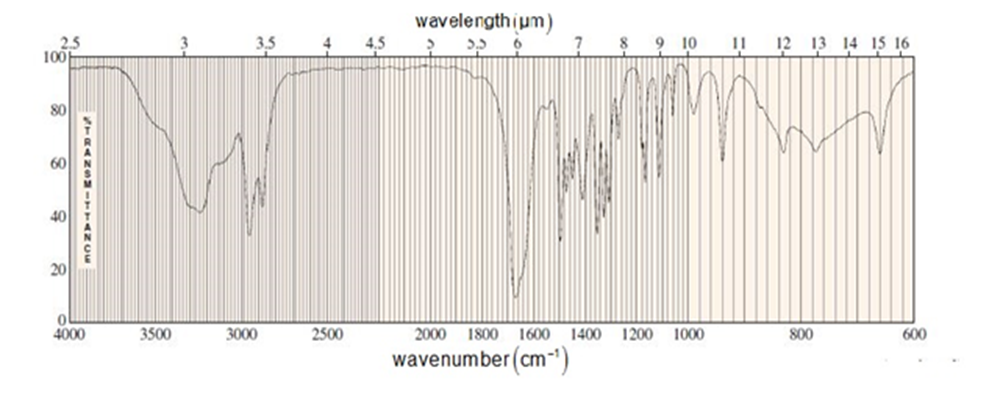
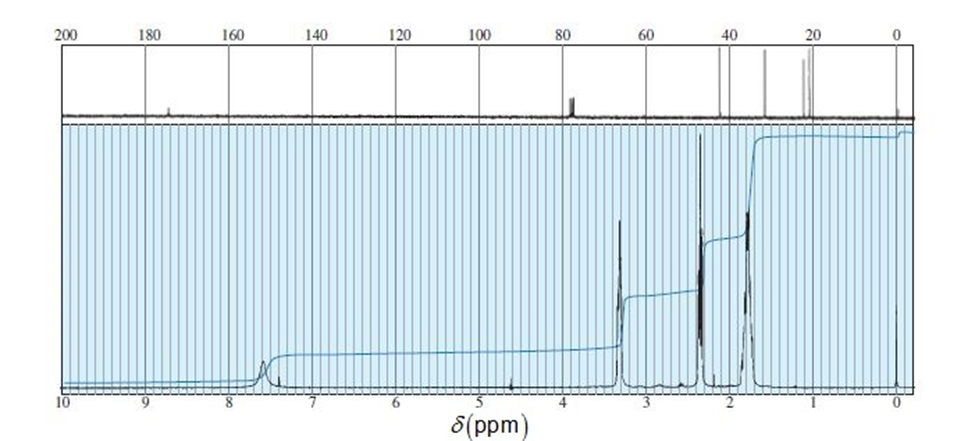
An unknown compound of the molecular formula C5H9NO gives the IR and NMR spectra shown here. The broad NMR peak at d 7.55 disappears when the sample is shaken with D2O . Propose a structure, and show how it is consistent with the absorptions in the spectra.
One mole of acetyl chloride is added to a liter of triethylamine, resulting in a vigorous exothermic reaction. Once the reaction mixture has cooled, 1 mole of ethanol is added. Another vigorous exothermic reaction results. The mixture is analyzed and found to contain triethylamine, ethyl acetate, and triethylammonium chloride. Propose mechanisms for the two exothermic reactions.
Show how you would accomplish the following syntheses using amides as intermediates.
You may use any necessary reagents.
What do you think about this solution?
We value your feedback to improve our textbook solutions.