Chapter 23: Q11P (page 1218)
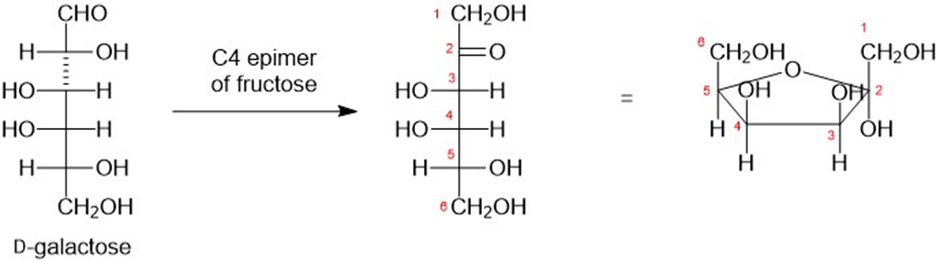
The carbonyl group in D-galactose may be isomerized from C1 to C2 by brief treatment with dilute base (by enediol rearrangement, Section 23-7). The product is the C4 epimer of fructose. Draw the furanose structure of the product.
Short Answer
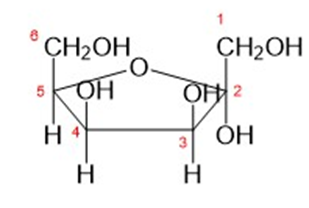
furanose structure of the product