Chapter 16: Q20P (page 823)
Borazole,  ,is an unusually stable cyclic compound. Propose a structure for borazole, and explain why it is aromatic.
,is an unusually stable cyclic compound. Propose a structure for borazole, and explain why it is aromatic.
Short Answer
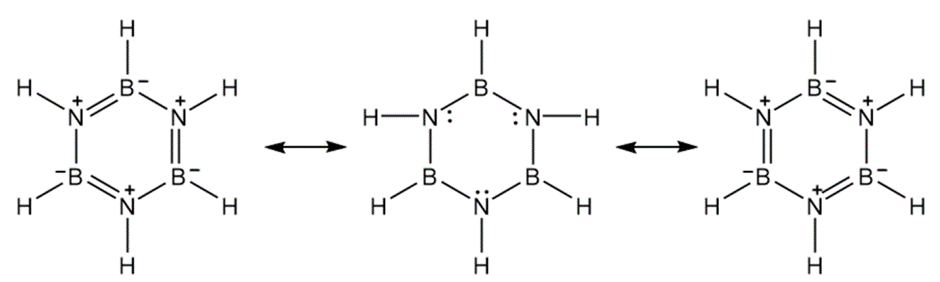
Resonance structure of borazole
Learning Materials
Features
Discover
Chapter 16: Q20P (page 823)
Borazole,  ,is an unusually stable cyclic compound. Propose a structure for borazole, and explain why it is aromatic.
,is an unusually stable cyclic compound. Propose a structure for borazole, and explain why it is aromatic.

Resonance structure of borazole
All the tools & learning materials you need for study success - in one app.
Get started for free
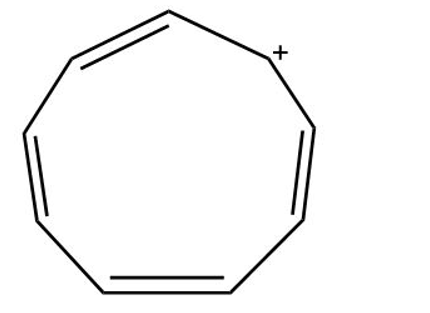
The following hydrocarbon has an unusually large dipole moment. Explain how a large dipole moment might arise
(a)Cyclopnonatetraene cation
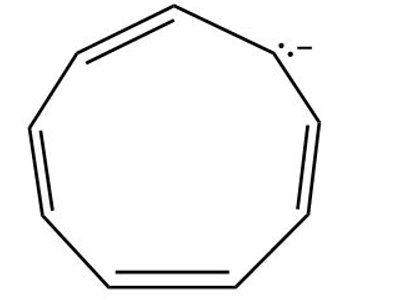
(b) Cyclononatetraene anion
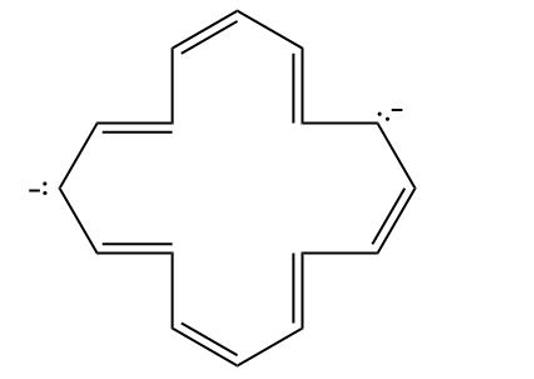
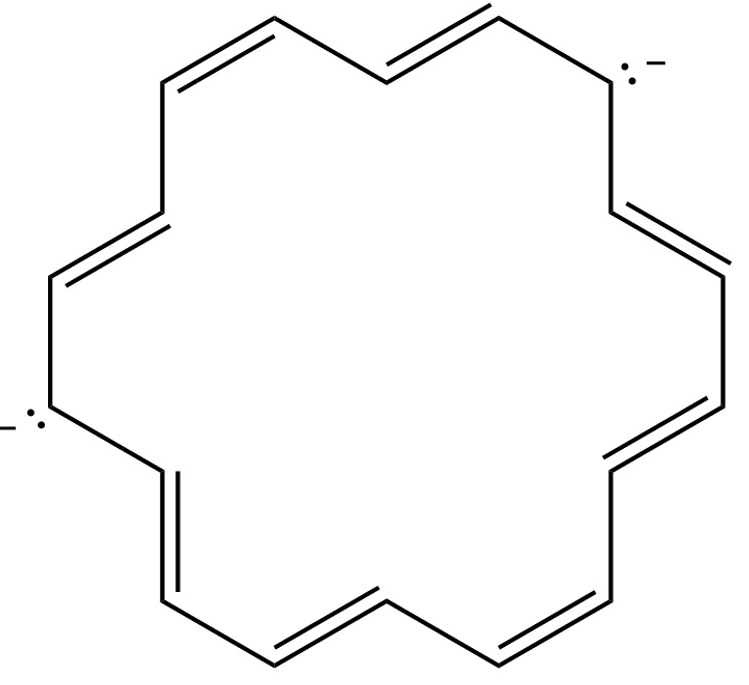
(c) [16] annulene dianion
(d) [18] annulene dianion
(e)
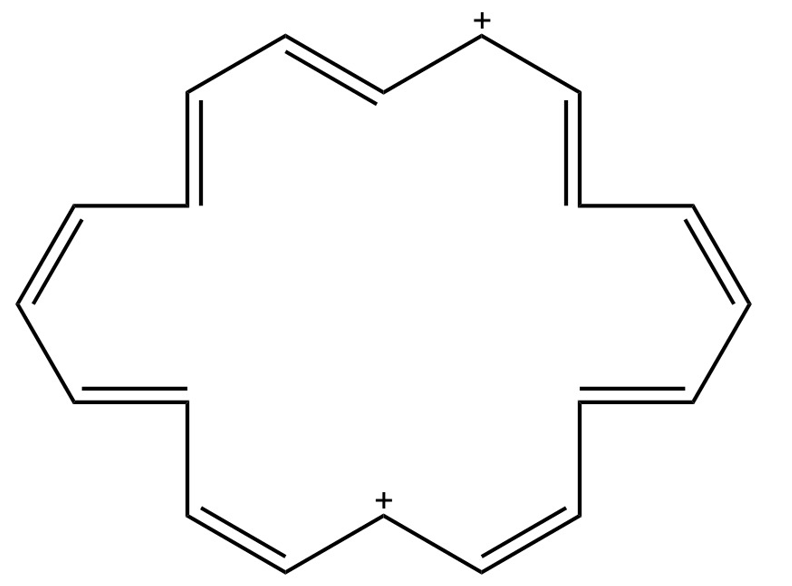
(f) [20] annulene dication
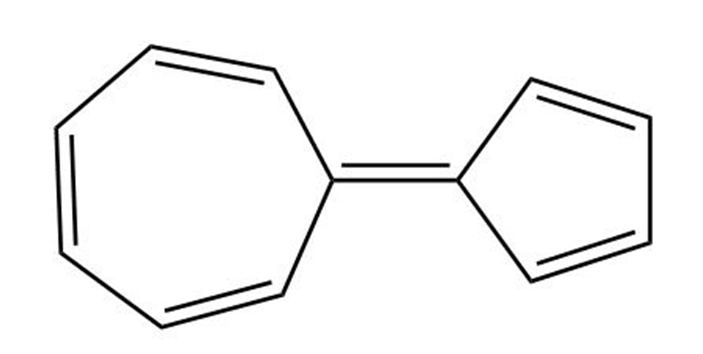
The following hydrocarbon has an unusually large dipole moment. Explain how a large dipole moment might arise.
Make a model of cyclooctatetraene in the tub conformation. Draw this conformation, and estimate the angle between the p orbitals of adjacent pi bonds
Ciprofloxacin is a member of the fluoroquinolone class of antibiotics.
(a) Which of its rings are aromatic?
(b) Which nitrogen atoms are basic?
(c) Which protons would you expect to appear between d 6 and d 8 in the proton NMR spectrum?
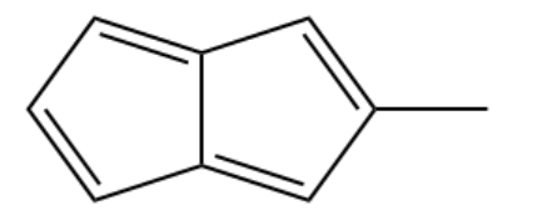
One of the following compounds is much more stable than the other two. Classify each as aromatic, antiaromatic, or nonaromatic
(a)Methyl heptalene
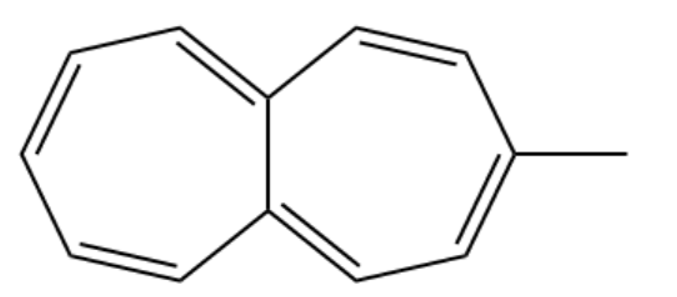
(b) Methyl azulene
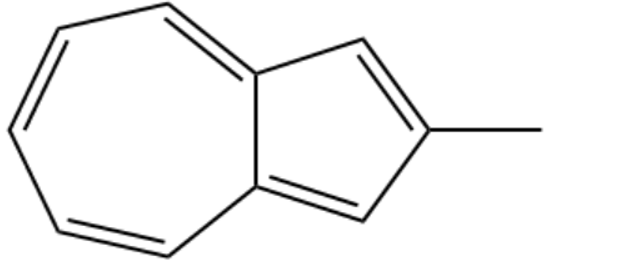
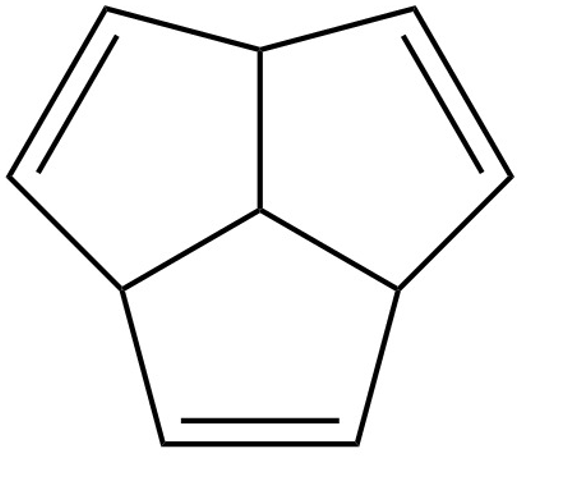
(c) Methyl pentalene
What do you think about this solution?
We value your feedback to improve our textbook solutions.