Chapter 16: Q13P (page 817)
The following hydrocarbon has an unusually large dipole moment. Explain how a large dipole moment might arise.

Short Answer
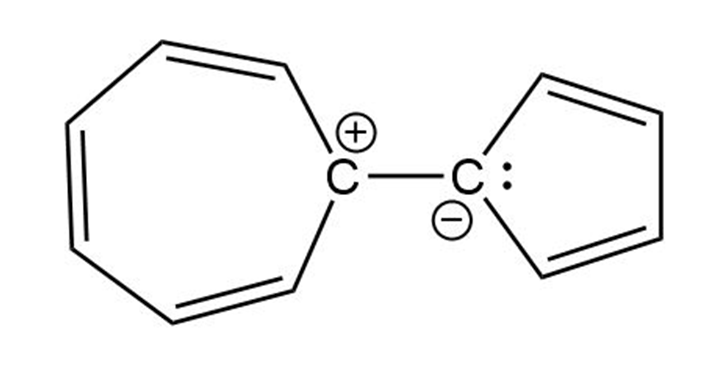
Charge separation (both rings are aromatic)
Learning Materials
Features
Discover
Chapter 16: Q13P (page 817)
The following hydrocarbon has an unusually large dipole moment. Explain how a large dipole moment might arise.


Charge separation (both rings are aromatic)
All the tools & learning materials you need for study success - in one app.
Get started for free
Ciprofloxacin is a member of the fluoroquinolone class of antibiotics.
(a) Which of its rings are aromatic?
(b) Which nitrogen atoms are basic?
(c) Which protons would you expect to appear between d 6 and d 8 in the proton NMR spectrum?
Question: The benzene ring alters the reactivity of a neighboring group in the benzylic position much as a double bond alters the reactivity of groups in the allylic position.
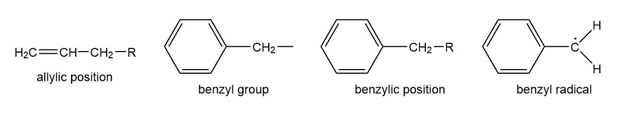
Benzylic cations, anions, and radicals are all more stable than simple alkyl intermediates.
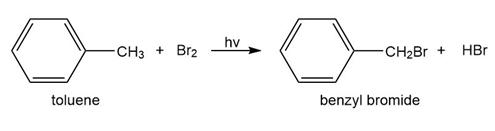
c. Which of the following reactions will have the faster rate and give the better yield? Use a drawing of the transition state to explain your answer.

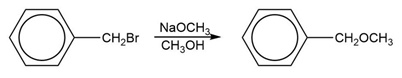
Does the MO energy diagram of cyclooctatetraene appear to be a particularly stable or unstable configuration? Explain
Question: For each NMR spectrum, propose a structure consistent with the spectrum and the additional information provided.
a. Elemental analysis shows the molecular formula to be C8H7OCl . The IR spectrum shows a moderate absorption at 1602 cm-1 and a strong absorption at 1690 cm-1 .
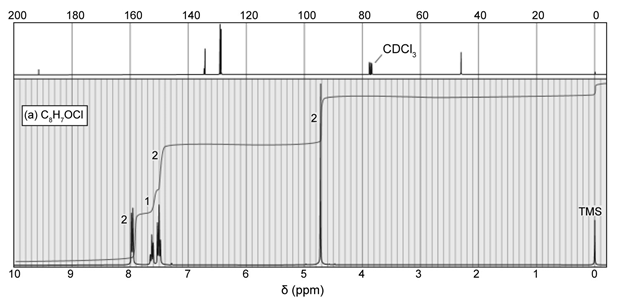
b. The mass spectrum shows a double molecular ion of ratio 1:1 at m/z 184 and 186.

Repeat Problem 16-10 for the cyclopentadienyl ions.
(a) Draw one all-bonding MO, then a pair of degenerate MOs, and then a final pair of degenerate MOs.
(b)Draw the energy diagram, fill in the electrons, and confirm the electronic configurations of the cyclopentadienyl cation and anion.
(c) Add electrons to your energy diagram to show the configuration of the cyclopentadienyl cation and the cyclopentadienyl anion. Which is aromatic and which is antiaromatic?
What do you think about this solution?
We value your feedback to improve our textbook solutions.