Chapter 16: 16-5P (page 809)
Does the MO energy diagram of cyclooctatetraene appear to be a particularly stable or unstable configuration? Explain
Short Answer
Learning Materials
Features
Discover
Chapter 16: 16-5P (page 809)
Does the MO energy diagram of cyclooctatetraene appear to be a particularly stable or unstable configuration? Explain

All the tools & learning materials you need for study success - in one app.
Get started for free
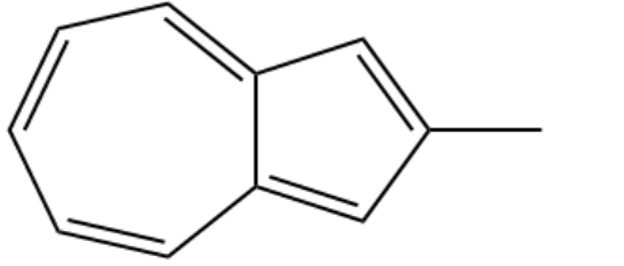
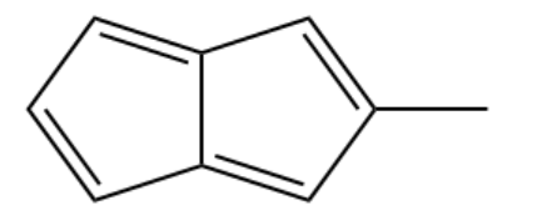
One of the following compounds is much more stable than the other two. Classify each as aromatic, antiaromatic, or nonaromatic
(a)Methyl heptalene
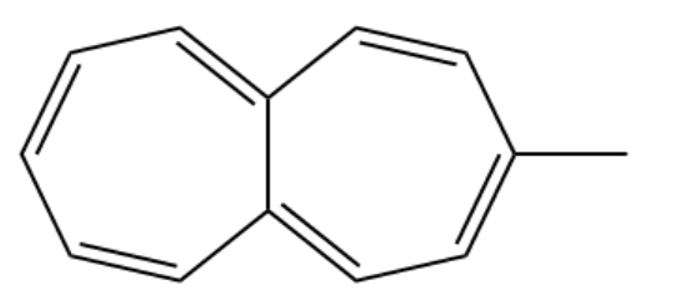
(b) Methyl azulene
(c) Methyl pentalene
Question: The benzene ring alters the reactivity of a neighboring group in the benzylic position much as a double bond alters the reactivity of groups in the allylic position.
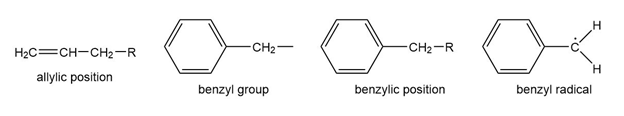
Benzylic cations, anions, and radicals are all more stable than simple alkyl intermediates.
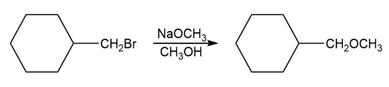
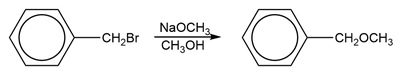
c. Which of the following reactions will have the faster rate and give the better yield? Use a drawing of the transition state to explain your answer.
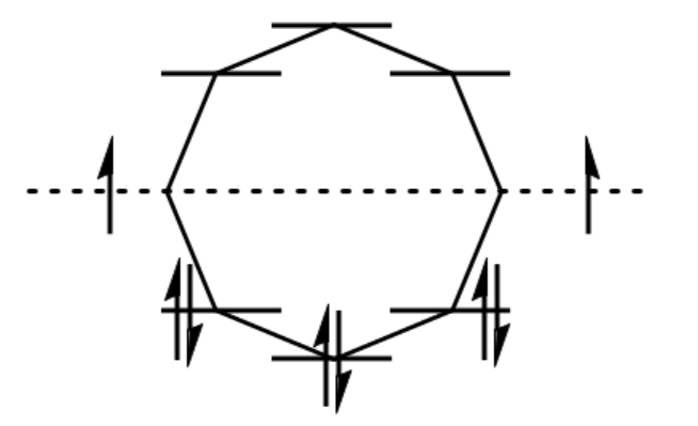
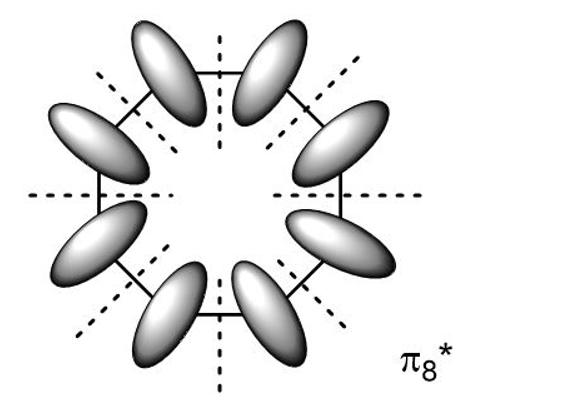
(a) Use the polygon rule to draw an energy diagram for the MOs of a planar cyclooctatetraenyl system.
(b) Fill in the eight pi electrons for cyclooctatetraene. Is this electronic configuration aromatic or antiaromatic? Could the cyclooctatetraene system be aromatic if it gained or lost electrons?
(c) Draw pictorial representations for the three bonding MOs and the two nonbonding MOs of cyclooctatetraene. The antibonding MOs are difficult to draw, except for the all-antibonding MO
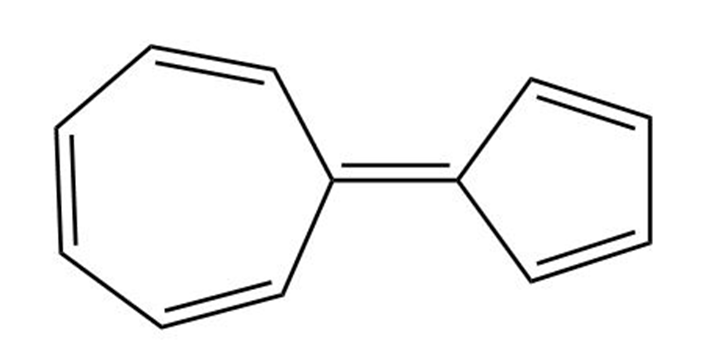
The following hydrocarbon has an unusually large dipole moment. Explain how a large dipole moment might arise.
Question: The benzene ring alters the reactivity of a neighboring group in the benzylic position much as a double bond alters the reactivity of groups in the allylic position.
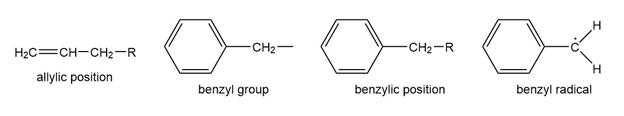
Benzylic cations, anions, and radicals are all more stable than simple alkyl intermediates.
a. Use resonance forms to show the delocalization (over four carbon atoms) of the positive charge, unpaired electron, and negative charge of the benzyl cation, radical, and anion.
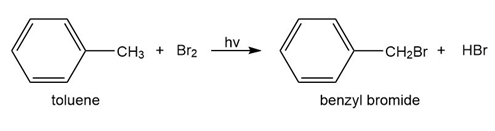
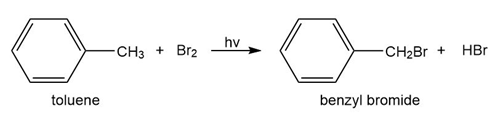
b. Toulene reacts with bromine in the presence of light to give benzyl bromide. Propose a mechanism for this reaction.
c. Which of the following reactions will have the faster rate and give the better yield? Use a drawing of the transition state to explain your answer.
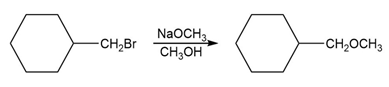
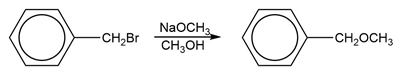
What do you think about this solution?
We value your feedback to improve our textbook solutions.