Chapter 16: 16-6P (page 811)
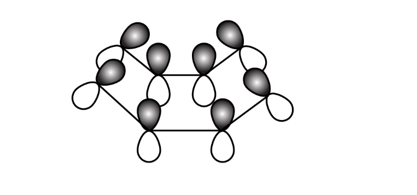
Make a model of cyclooctatetraene in the tub conformation. Draw this conformation, and estimate the angle between the p orbitals of adjacent pi bonds
Short Answer
Learning Materials
Features
Discover
Chapter 16: 16-6P (page 811)
Make a model of cyclooctatetraene in the tub conformation. Draw this conformation, and estimate the angle between the p orbitals of adjacent pi bonds

All the tools & learning materials you need for study success - in one app.
Get started for free
Does the MO energy diagram of cyclooctatetraene appear to be a particularly stable or unstable configuration? Explain
(a) Draw all the Kekulé structures of anthracene and phenanthrene.
(b) Propose mechanisms for the two additions shown.
(c) In Chapter 8, most of the additions of bromine to double bonds gave entirely anti stereochemistry. Explain why the addition to phenanthrene gives a mixture of syn andanti stereochemistry.
(d) When the product from (c) is heated, HBr is evolved, and 9-bromophenanthrene results. Propose a mechanism for this dehydrohalogenation.
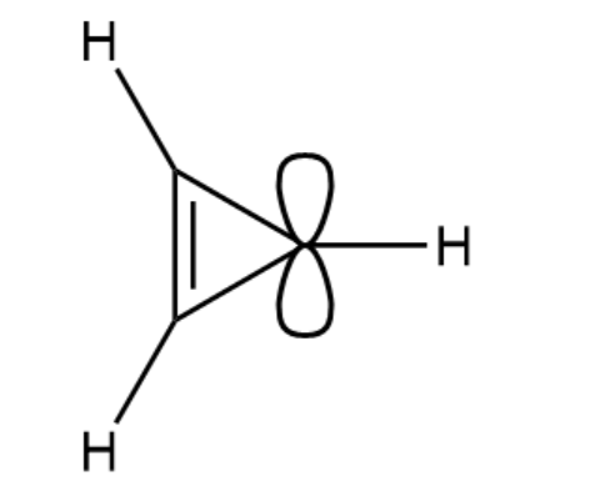
(a) Draw the molecular orbitals for the cyclopropenyl case. H H H (Because there are three p orbitals, there must be three MOs: one all-bonding MO and one degenerate pair of MOs.)
(b) Draw an energy diagram for the cyclopropenyl MOs. (The polygon rule is helpful.) Label each MO as bonding, nonbonding, or antibonding, and add the nonbonding line. Notice that it goes through the approximate average of the MOs.
(c) Add electrons to your energy diagram to show the configuration of the cyclopropenyl cation and the cyclopropenyl anion. Which is aromatic and which is antiaromatic?
Question: The benzene ring alters the reactivity of a neighboring group in the benzylic position much as a double bond alters the reactivity of groups in the allylic position.
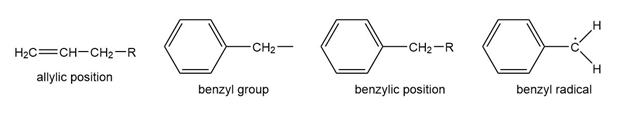
Benzylic cations, anions, and radicals are all more stable than simple alkyl intermediates.
a. Use resonance forms to show the delocalization (over four carbon atoms) of the positive charge, unpaired electron, and negative charge of the benzyl cation, radical, and anion.
b. Toulene reacts with bromine in the presence of light to give benzyl bromide. Propose a mechanism for this reaction.
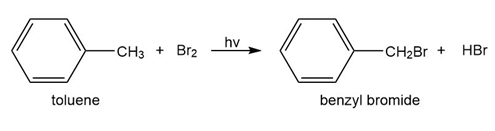
c. Which of the following reactions will have the faster rate and give the better yield? Use a drawing of the transition state to explain your answer.
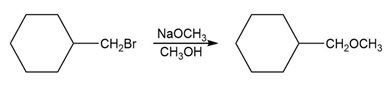
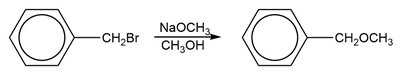
When 3-chlorocyclopropene is treated with AgBF4, AgCl precipitates. The organic product can be obtained as a crystalline material, soluble in polar solvents such as nitromethane but insoluble in hexane. When the crystalline material is dissolved in nitromethane containing KCl, the original 3-chlorocyclopropene is regenerated. Determine the structure of the crystalline material, and write equations for its formation and its reaction with chloride ion
What do you think about this solution?
We value your feedback to improve our textbook solutions.