Chapter 16: 16-7P (page 812)
Classify the following compounds as aromatic, antiaromatic, or nonaromatic
(a)
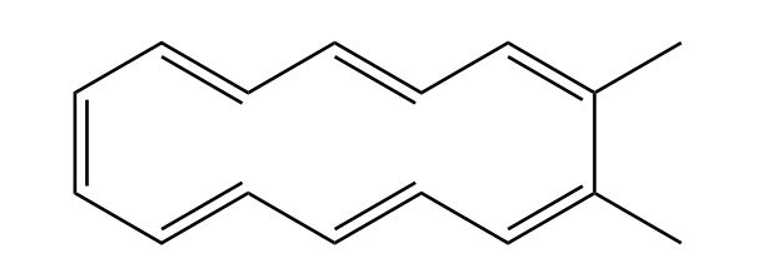
(b)
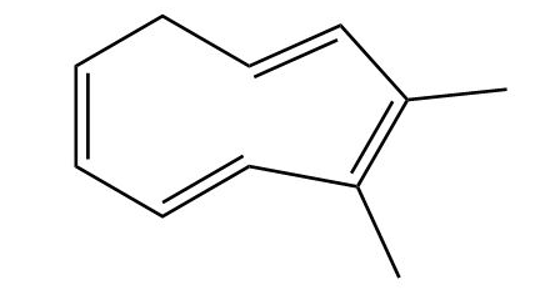
(c)
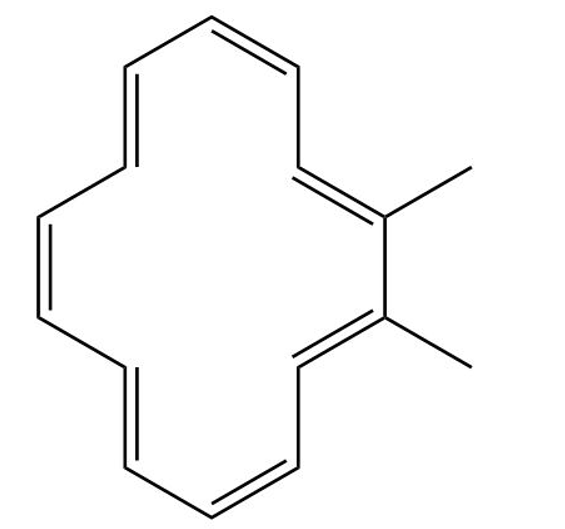
(d)
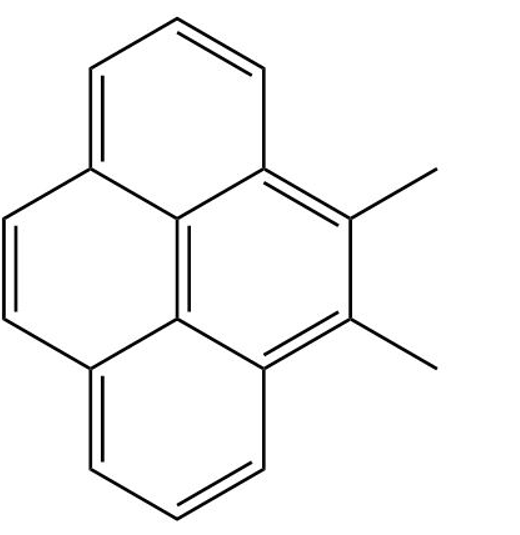
Short Answer
(a)
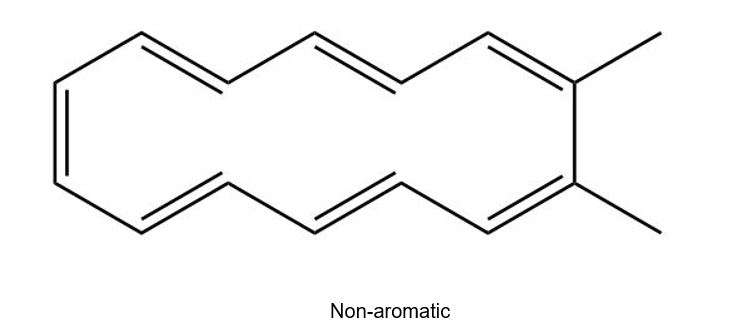
(b)
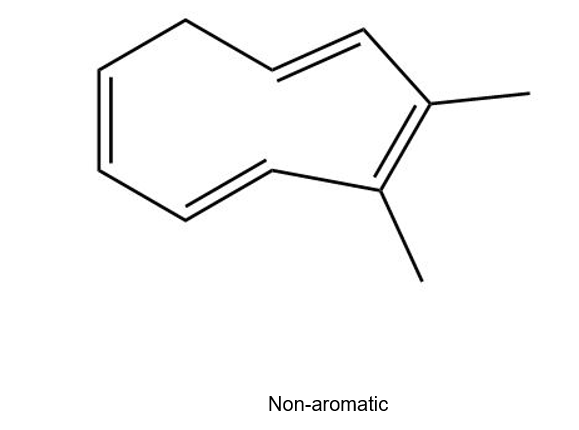
(c)
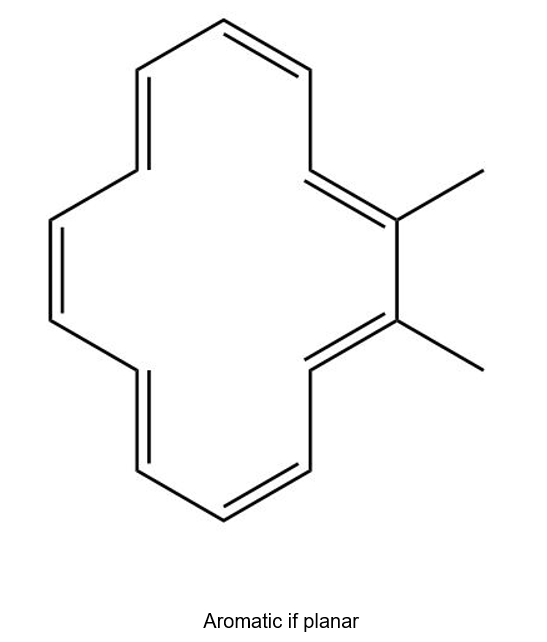
(d)
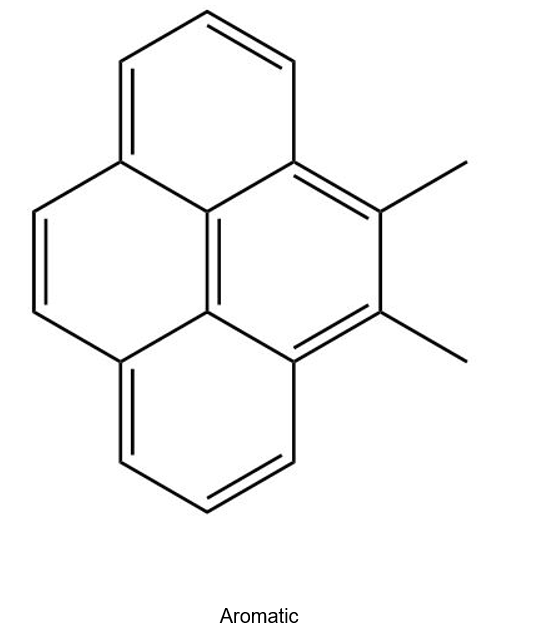
Learning Materials
Features
Discover
Chapter 16: 16-7P (page 812)
Classify the following compounds as aromatic, antiaromatic, or nonaromatic
(a)

(b)

(c)

(d)

(a)

(b)

(c)

(d)

All the tools & learning materials you need for study success - in one app.
Get started for free
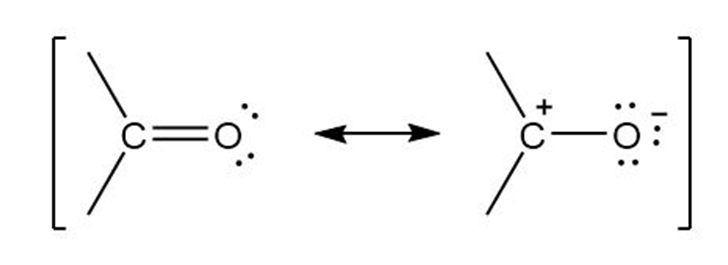
The polarization of a carbonyl group can be represented by a pair of resonance structures
Cyclopropenone and cycloheptatrienone are more stable than anticipated. Cyclopentadienone, however, is relatively unstable and rapidly undergoes a Diels–Alder dimerization. Explain.
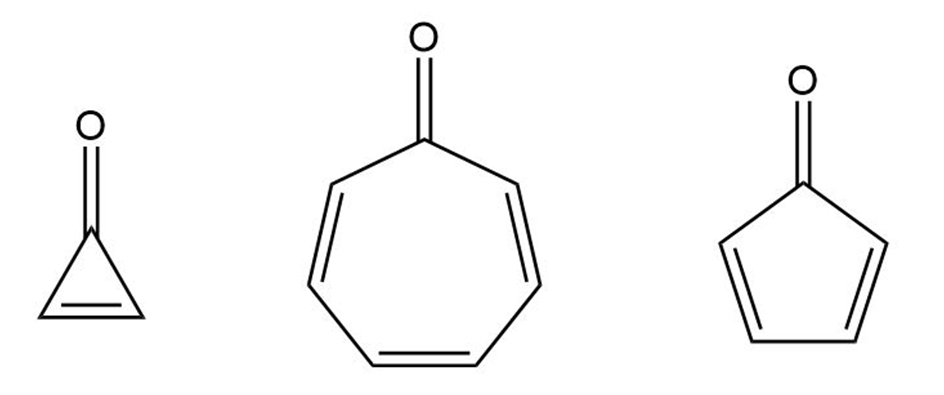
Cyclopropenone CycloheptatrienoneCyclopentadienone
Borazole,  ,is an unusually stable cyclic compound. Propose a structure for borazole, and explain why it is aromatic.
,is an unusually stable cyclic compound. Propose a structure for borazole, and explain why it is aromatic.
Explain why each compound is aromatic, antiaromatic, or nonaromatic
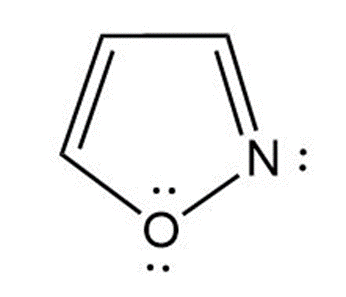
(a) Isoxazole
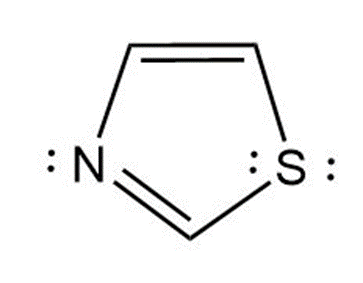
(b) 1,3-thiazole
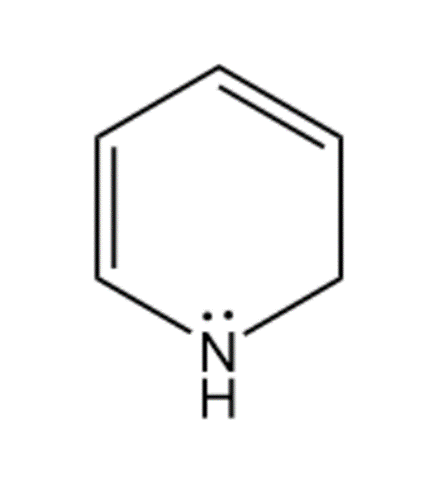
(c) Pyran
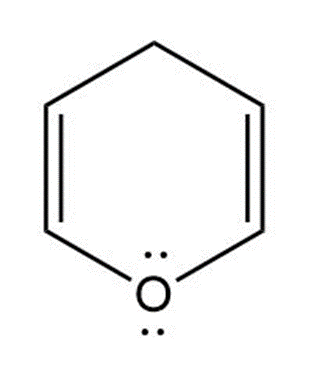
(d) Pyrylium ion
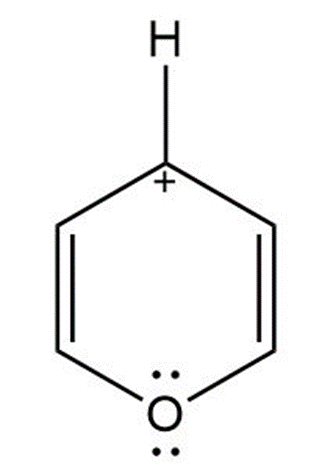
(e) γ - pyrone
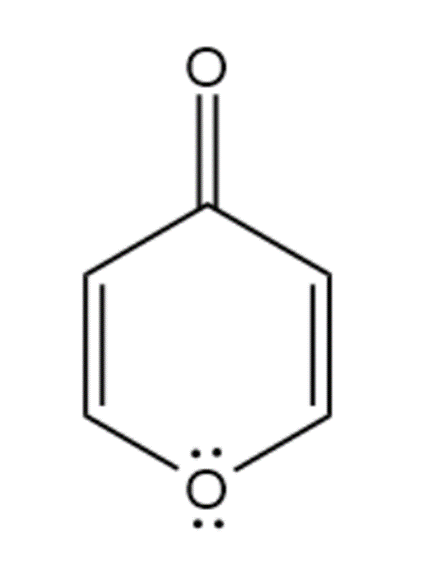
(f) 1,2-Dihydropyridine
(g)Cytosine
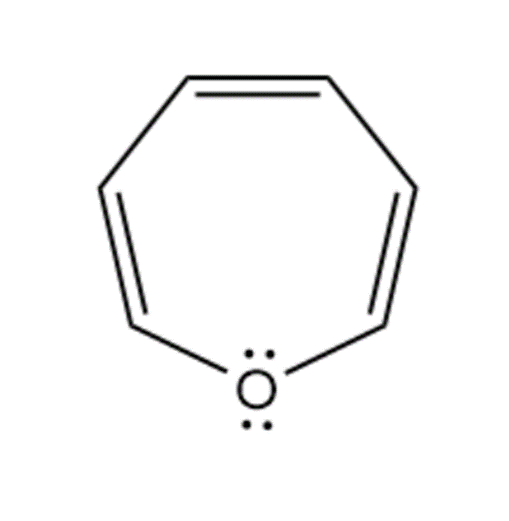
(h)
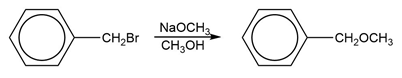
Question: The benzene ring alters the reactivity of a neighboring group in the benzylic position much as a double bond alters the reactivity of groups in the allylic position.
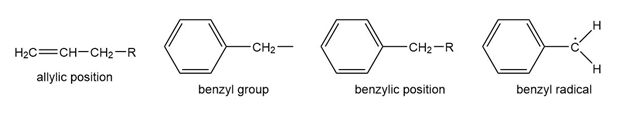
Benzylic cations, anions, and radicals are all more stable than simple alkyl intermediates.
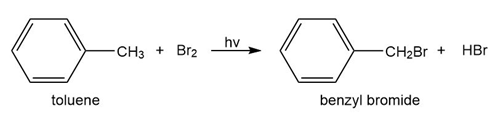
c. Which of the following reactions will have the faster rate and give the better yield? Use a drawing of the transition state to explain your answer.
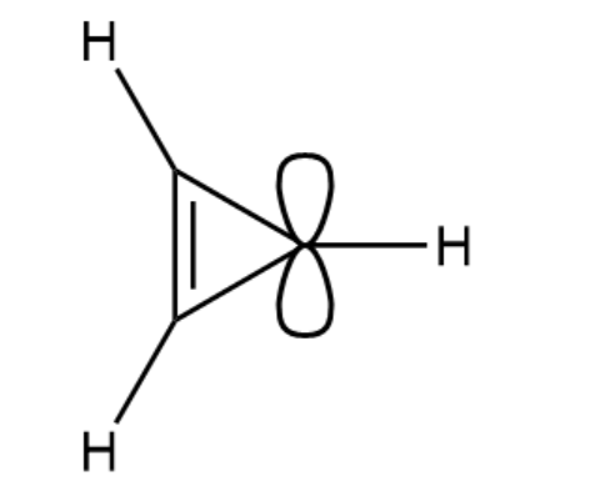
(a) Draw the molecular orbitals for the cyclopropenyl case. H H H (Because there are three p orbitals, there must be three MOs: one all-bonding MO and one degenerate pair of MOs.)
(b) Draw an energy diagram for the cyclopropenyl MOs. (The polygon rule is helpful.) Label each MO as bonding, nonbonding, or antibonding, and add the nonbonding line. Notice that it goes through the approximate average of the MOs.
(c) Add electrons to your energy diagram to show the configuration of the cyclopropenyl cation and the cyclopropenyl anion. Which is aromatic and which is antiaromatic?
What do you think about this solution?
We value your feedback to improve our textbook solutions.