Chapter 16: 16-4P (page 805)
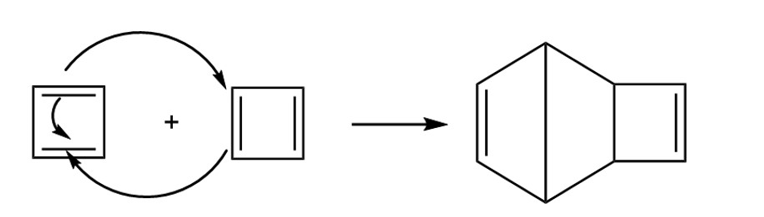
Show the product of the Diels–Alder dimerization of cyclobutadiene.
Short Answer
Learning Materials
Features
Discover
Chapter 16: 16-4P (page 805)
Show the product of the Diels–Alder dimerization of cyclobutadiene.

All the tools & learning materials you need for study success - in one app.
Get started for free
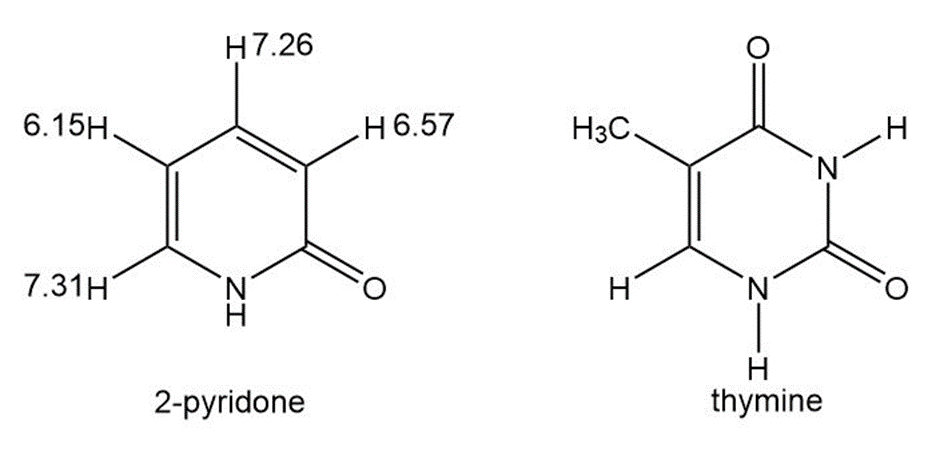
The proton NMR spectrum of 2-pyridone gives the chemical shifts shown.
(a) Is 2-pyridone aromatic?
(b) Use resonance forms to explain your answer to (a). Also explain why the protons at d 7.31 and d 7.26 are more deshielded than the other two (d 6.15 and d 6.57).
(c) Thymine is one of the heterocyclic bases found in DNA. Do you expect thymine to be aromatic? Explain.
(d) The structure of 5-fluorouracil is shown in the box at the side of the page. Is 5-fluorouracil aromatic? Explain.
Does the MO energy diagram of cyclooctatetraene appear to be a particularly stable or unstable configuration? Explain
Ciprofloxacin is a member of the fluoroquinolone class of antibiotics.
(a) Which of its rings are aromatic?
(b) Which nitrogen atoms are basic?
(c) Which protons would you expect to appear between d 6 and d 8 in the proton NMR spectrum?
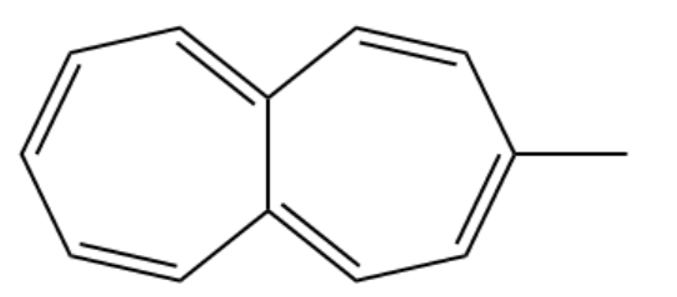
One of the following compounds is much more stable than the other two. Classify each as aromatic, antiaromatic, or nonaromatic
(a)Methyl heptalene
(b) Methyl azulene
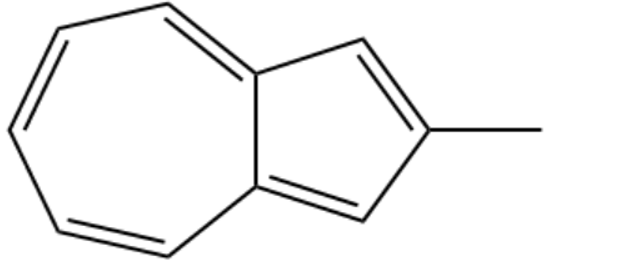
(c) Methyl pentalene
When 3-chlorocyclopropene is treated with AgBF4, AgCl precipitates. The organic product can be obtained as a crystalline material, soluble in polar solvents such as nitromethane but insoluble in hexane. When the crystalline material is dissolved in nitromethane containing KCl, the original 3-chlorocyclopropene is regenerated. Determine the structure of the crystalline material, and write equations for its formation and its reaction with chloride ion
What do you think about this solution?
We value your feedback to improve our textbook solutions.