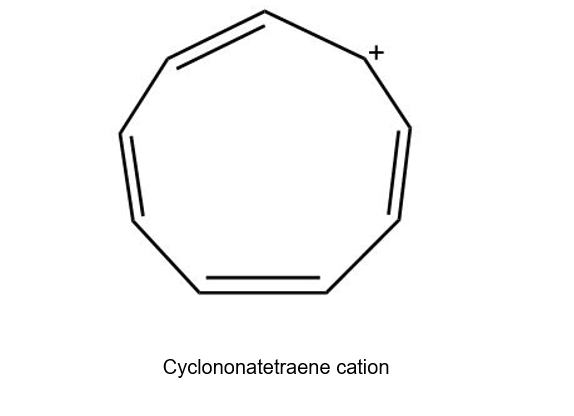
(a) Cyclononatetraene cation is cyclic having continuous overlapping of p orbitals and it has 4n pi electrons. Hence, it is antiaromatic.
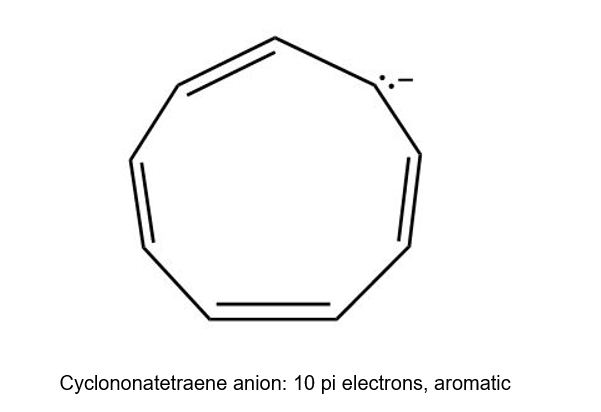
(b) Cyclononatetraene anion is cyclic having continuous overlapping of p orbital and it has 4n+2 pi electrons. Hence, it is aromatic.
(c) [16] annulene dianion is cyclic having continuous overlapping of p orbital and it has 4n+2 pi electrons. Hence, it is aromatic.
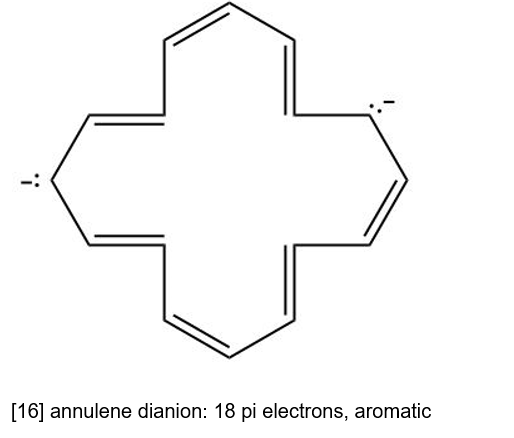
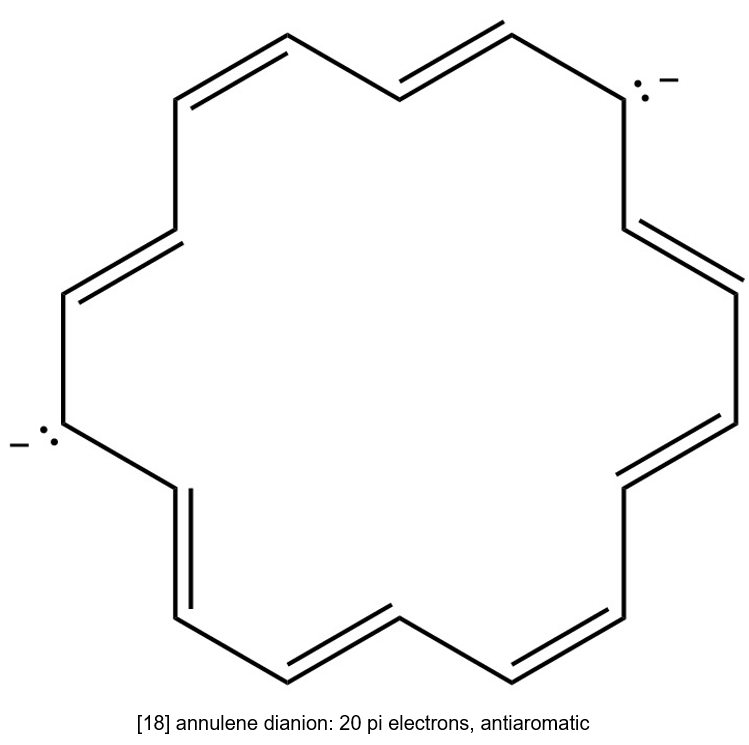
(d) [18] annulene dianion is cyclic having continuous overlapping of p orbital and it has 4n pi electrons. Hence, it is antiaromatic.
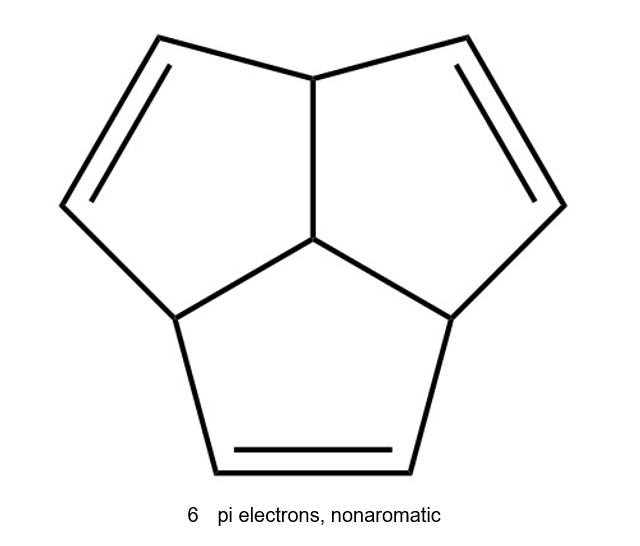
(e) The given compound is cyclic but does not have a continuous, overlapping ring of p orbitals. Hence, it is non aromatic.
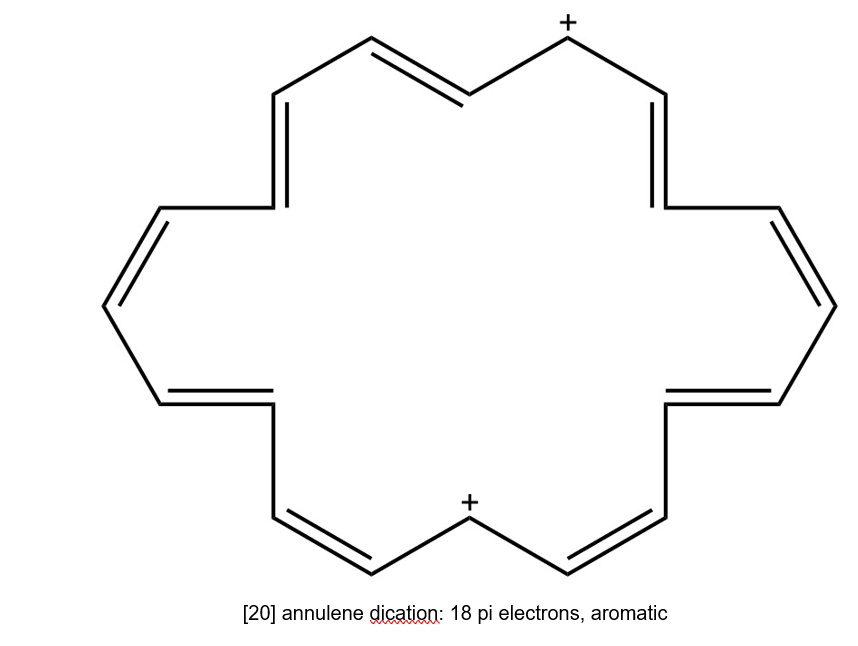
(f) [20] annulene dication is cyclic having continuous overlapping of p orbital and it has 4n+2 pi electrons. Hence, it is aromatic.