Chapter 24: Q7P (page 1267)
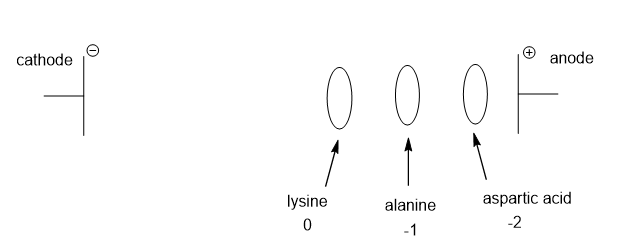
Draw the electrophoretic separation of Ala, Lys, and Asp at pH 9.7.
Short Answer
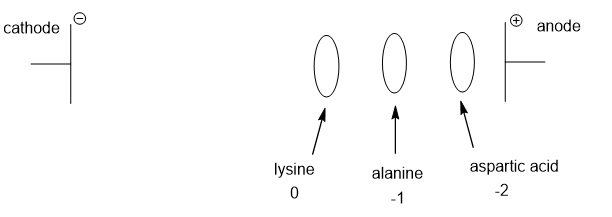
At pH 9.7, alanine has isoelectric point 6.0, and has a charge of -1, hence will migrate towards anode. Lysine which has isoelectric point of 9.7 will not move and aspartic acid having isoelectric point of 2.8 has a charge of -2 and will also migrate towards the anode faster than alanine.