Chapter 24: Q25P (page 1289)
Show how you would synthesize Leu-Gly-Ala-Val-Phe starting with Fmoc-Ala-Val-Phe— P
Short Answer
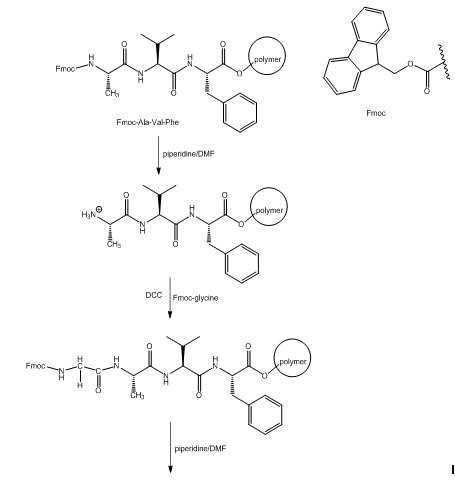
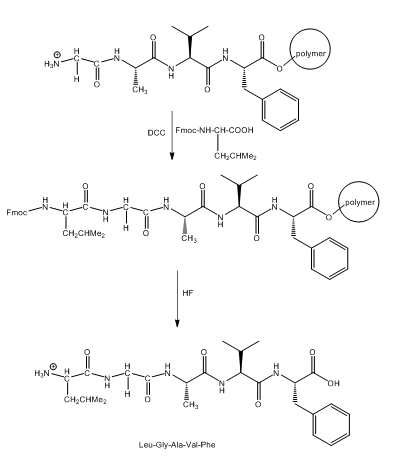
The synthesis of Leu-Gly-Ala-Val-Phe starting from Fmoc-Ala-Val-Phe-P is given as follows:
Learning Materials
Features
Discover
Chapter 24: Q25P (page 1289)
Show how you would synthesize Leu-Gly-Ala-Val-Phe starting with Fmoc-Ala-Val-Phe— P
The synthesis of Leu-Gly-Ala-Val-Phe starting from Fmoc-Ala-Val-Phe-P is given as follows:


All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the structure of the predominant form of
(a)Isoleucine at pH 11 (b) Proline at pH 2
(c)Arginine at pH 7 (d) Glutamic acid at pH 7
A mixture of alanine, lysine, and aspartic acid at 1). pH 6 ; 2). pH 11; 3). pH 2
Complete hydrolysis of an unknown basic decapeptide gives Gly, Ala, Leu, Ile, Phe, Tyr, Glu, Arg, Lys, and Ser. Terminal residue analysis shows that the N terminus is Ala, and the C terminus is Ile. Incubation of the decapeptide with chymotrypsin gives two tripeptides, A and B, and a tetrapeptide, C. Amino acid analysis shows that peptide A contains Gly, Glu, Tyr, and; peptide B contains Ala, Phe, and Lys; and peptide C contains Leu, Ile, Ser, and Arg.Terminal residue analysis gives the following results.

Incubation of the decapeptide with trypsin gives a dipeptide D, a pentapeptide E, and a tripeptide F. Terminal residue analysis of F shows that the N terminus is Ser and the C terminus is Ile. Propose a structure for the decapeptide and for fragments A through F.
Show the steps and intermediates in the synthesis of Leu-Ala-Phe by the solid-phase process.
There are many methods for activating a carboxylic acid in preparation for coupling with an amine. The following method converts the acid to an N-hydroxysuccinimide (NHS) ester.
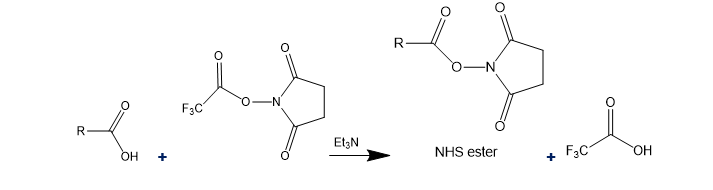
(a) Explain why an NHS ester is much more reactive than a simple alkyl ester.
(b) Propose a mechanism for the reaction shown.
(c) Propose a mechanism for the reaction of the NHS ester with an amine, R-NH2
Draw the complete structures of the following peptides:
(a) Thr-Phe-Met (b) Serylarginylglycylphenylalanine (c) IMQDK (d) ELVIS
What do you think about this solution?
We value your feedback to improve our textbook solutions.