Chapter 24: Q26P (page 1289)
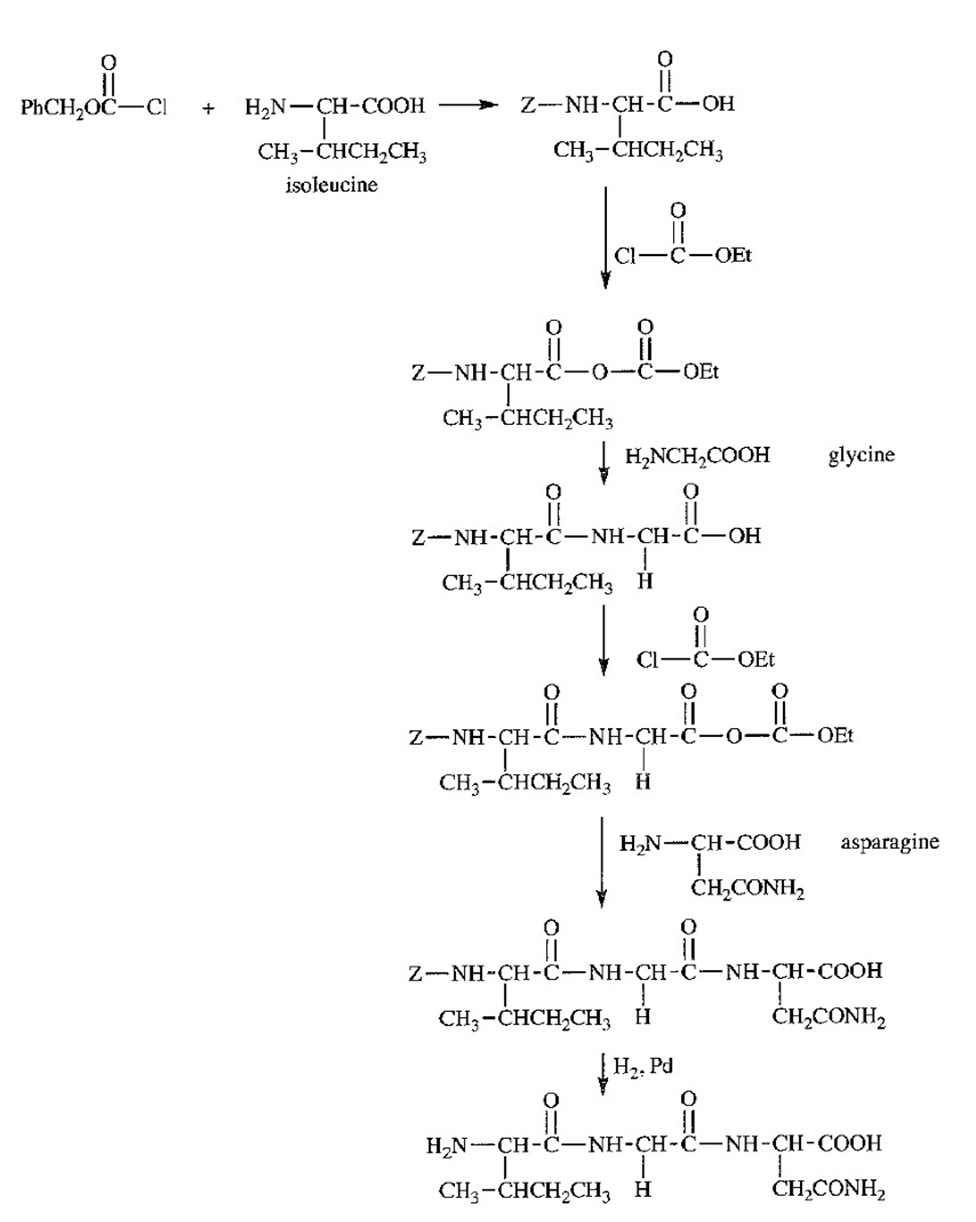
Show how solid-phase peptide synthesis would be used to make Ile-Gly-Asn.
Short Answer
Ile-Gly-Asn
Learning Materials
Features
Discover
Chapter 24: Q26P (page 1289)
Show how solid-phase peptide synthesis would be used to make Ile-Gly-Asn.

Ile-Gly-Asn
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the structure of the phenylthiohydantoin derivatives of
(a) Alanine (b) Tryptophan (c) Lysine (d) proline
The herbicide glyphosate(Roundup) kills plants by inhibiting an enzyme needed for synthesis of phenylalanine. Deprived of phenylalanine, the plant cannot make the proteins it needs, and it gradually weakens and dies. Although a small amount of glyphosate is deadly to a plant, its human toxicity is quite low. Suggest why this powerful herbicide has little effect on humans.
Lipoic acid is often found near the active sites of enzymes, usually bound to the peptide by a long, flexible amide linkage with a lysine residue.
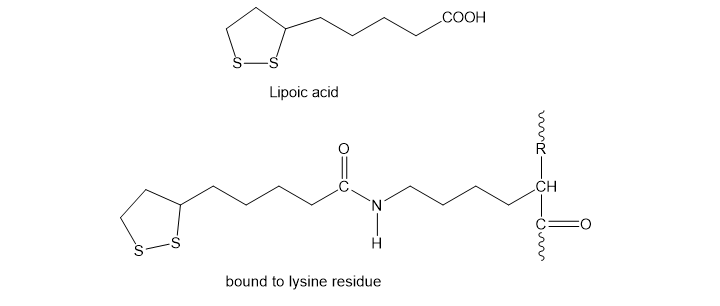
(a) Is lipoic acid a mild oxidizing agent or a mild reducing agent? Draw it in both its oxidized and reduced forms.
(b) Show how lipoic acid might react with two Cys residues to form a disulfide bridge.
(c) Give a balanced equation for the hypothetical oxidation or reduction, as you predicted in part (a), of an aldehyde by lipoic acid.
After treatment with peroxyformic acid, the peptide hormone vasopressin is partially hydrolyzed. The following fragments are recovered. Propose a structure for vasopressin.
Phe-Gln-Asn Pro-Arg-Gly.NH2 Cys-Tyr-Phe
Asn-Cys-Pro-Arg Tyr-Phe-Gln-Asn
Histidine is an important catalytic residue found at the active sites of many enzymes. In many cases, histidine appears to remove protons or to transfer protons from one location to another.
(a) Show which nitrogen atom of the histidine heterocycle is basic and which is not.
(b) Use resonance forms to show why the protonated form of histidine is a particularly stable cation.
(c) Show the structure that results when histidine accepts a proton on the basic nitrogen of the heterocycle and then is deprotonated on the other heterocyclic nitrogen. Explain how histidine might function as a pipeline to transfer protons between sites within an enzyme and its substrate.
What do you think about this solution?
We value your feedback to improve our textbook solutions.