Chapter 24: Q24P. (page 1287)
Question. Propose a mechanism for the coupling of acetic acid and aniline using DCC as a coupling agent.
Short Answer
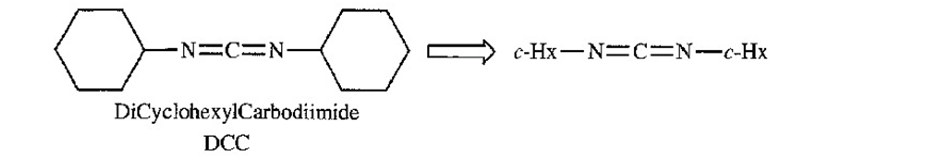
Representation of DCC as c-Hx

Mechanism for the coupling of acetic acid aniline
Learning Materials
Features
Discover
Chapter 24: Q24P. (page 1287)
Question. Propose a mechanism for the coupling of acetic acid and aniline using DCC as a coupling agent.

Representation of DCC as c-Hx

Mechanism for the coupling of acetic acid aniline
All the tools & learning materials you need for study success - in one app.
Get started for free
Aspartame(Nutrasweet®) is a remarkably sweet-tasting dipeptide ester. Complete hydrolysis of aspartame gives phenylalanine, aspartic acid, and methanol. Mild incubation with carboxypeptidase has no effect on aspartame. Treatment of aspartame with phenyl isothiocyanate, followed by mild hydrolysis, gives the phenylthiohydantoin of aspartic acid. Propose a structure for aspartame.
Write the complete structures for the following peptides. Tell whether each peptide is acidic, basic, or neutral.
Draw the complete structure of the following peptide Met-Ser-Gln-
Draw the structure of the phenylthiohydantoin derivatives of
(a) Alanine (b) Tryptophan (c) Lysine (d) proline
After treatment with peroxyformic acid, the peptide hormone vasopressin is partially hydrolyzed. The following fragments are recovered. Propose a structure for vasopressin.
Phe-Gln-Asn Pro-Arg-Gly.NH2 Cys-Tyr-Phe
Asn-Cys-Pro-Arg Tyr-Phe-Gln-Asn
What do you think about this solution?
We value your feedback to improve our textbook solutions.