The introduction of bromine at the position of a carboxylic acid is given by the Hell-Volhard-Zelensky reaction. In the first step of this reaction, a carboxylic acid is transformed into an Bromo-acid by using and . The second step of the reaction represents a reaction in which ammonia replaces bromine.
(a)
The synthesis of glycine by using bromination followed by amination is as shown below:
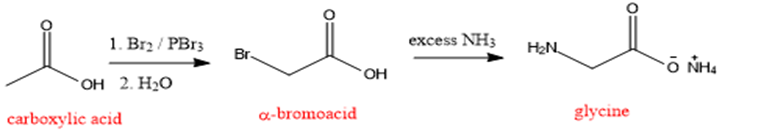
Formation of glycine
(b)
The synthesis of leucine by using bromination followed by amination is as shown below:
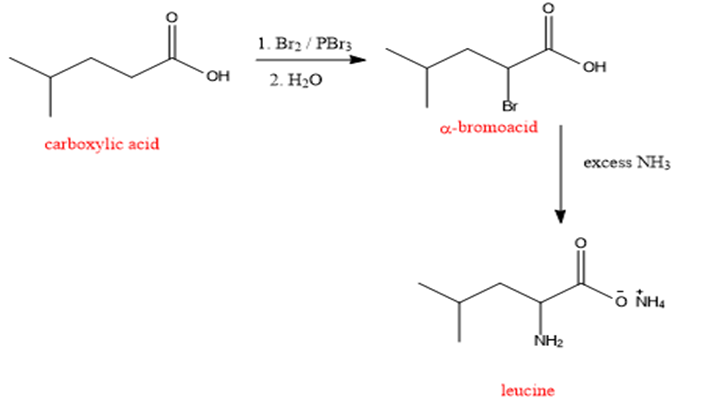
Formation of leucine
(c)
The synthesis of glutamic acid by using bromination followed by amination is as shown below:
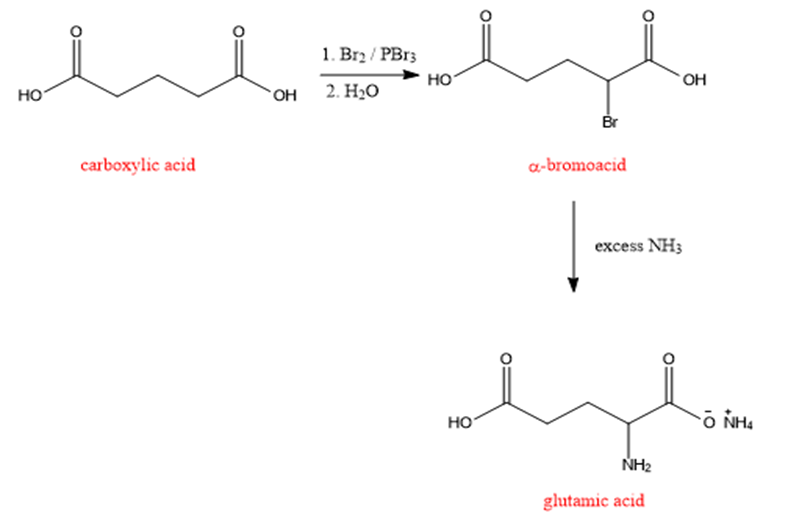
Formation of glutamic acid








