Chapter 24: Q17P. (page 1273)
Use resonance forms to show delocalization of the negative charge in Ruhemann’s purple anion.
Short Answer
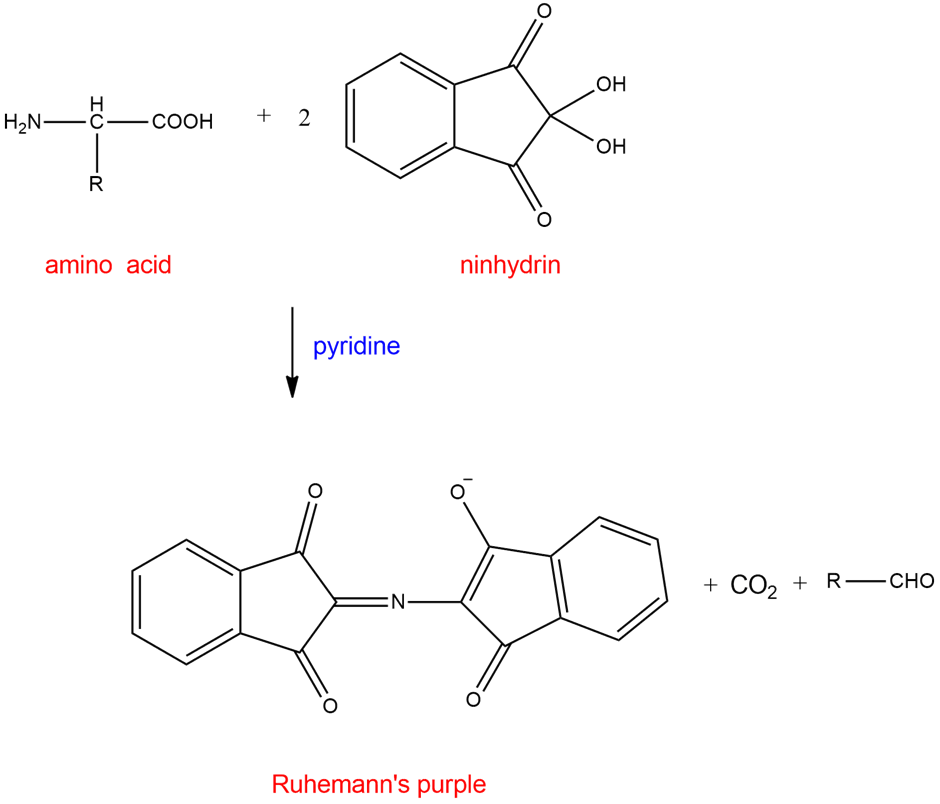
Formation of Ruhemann’s purple anion
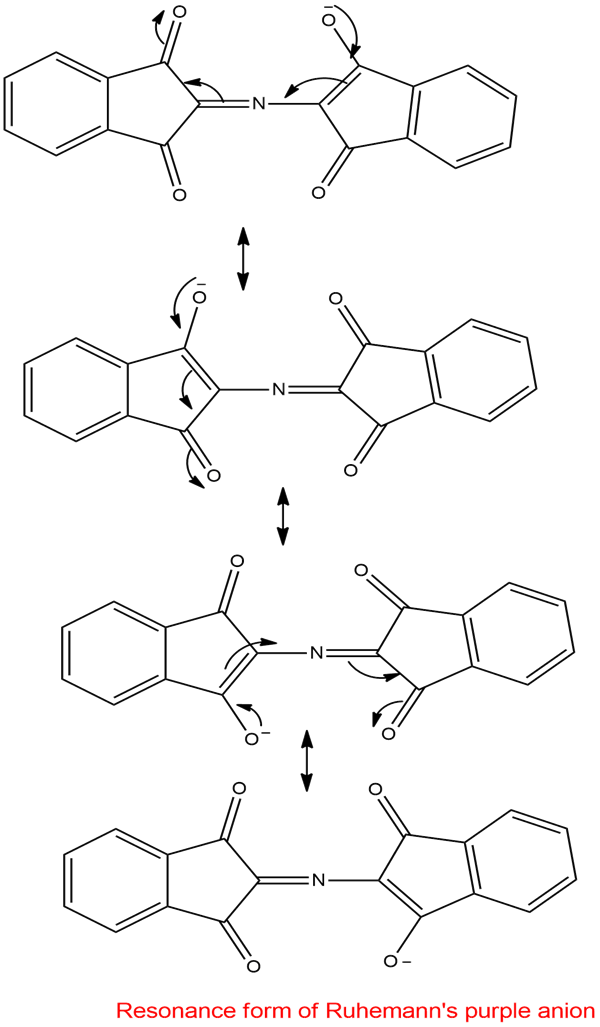
Resonance form of Ruhemann’s purple anion
Learning Materials
Features
Discover
Chapter 24: Q17P. (page 1273)
Use resonance forms to show delocalization of the negative charge in Ruhemann’s purple anion.

Formation of Ruhemann’s purple anion

Resonance form of Ruhemann’s purple anion
All the tools & learning materials you need for study success - in one app.
Get started for free
The following structure is drawn in an unconventional manner.
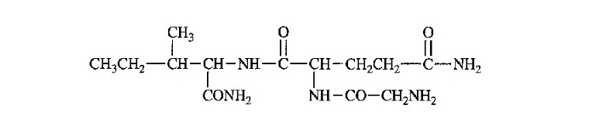
(a) Label the N terminus and the C terminus.
(b) Label the peptide bonds.
(c) Identify and label each amino acid present.
(d) Give the full name and the abbreviated name
Suggest a method for the synthesis of the natural L enantiomer of alanine from the readily available L enantiomer of lactic acid.

Show the steps and intermediates in the synthesis of Leu-Ala-Phe by the solid-phase process.
Draw the complete structures of the following peptides:
(a) Thr-Phe-Met (b) Serylarginylglycylphenylalanine (c) IMQDK (d) ELVIS
Question. Propose a mechanism for the coupling of acetic acid and aniline using DCC as a coupling agent.
What do you think about this solution?
We value your feedback to improve our textbook solutions.