Chapter 24: Q16P. (page 1273)
Give equations for the formation and hydrogenolysis of N-benzyloxy carbonyl methionine.
Short Answer
 Formation of N- benzyloxy carbonyl methionine
Formation of N- benzyloxy carbonyl methionine
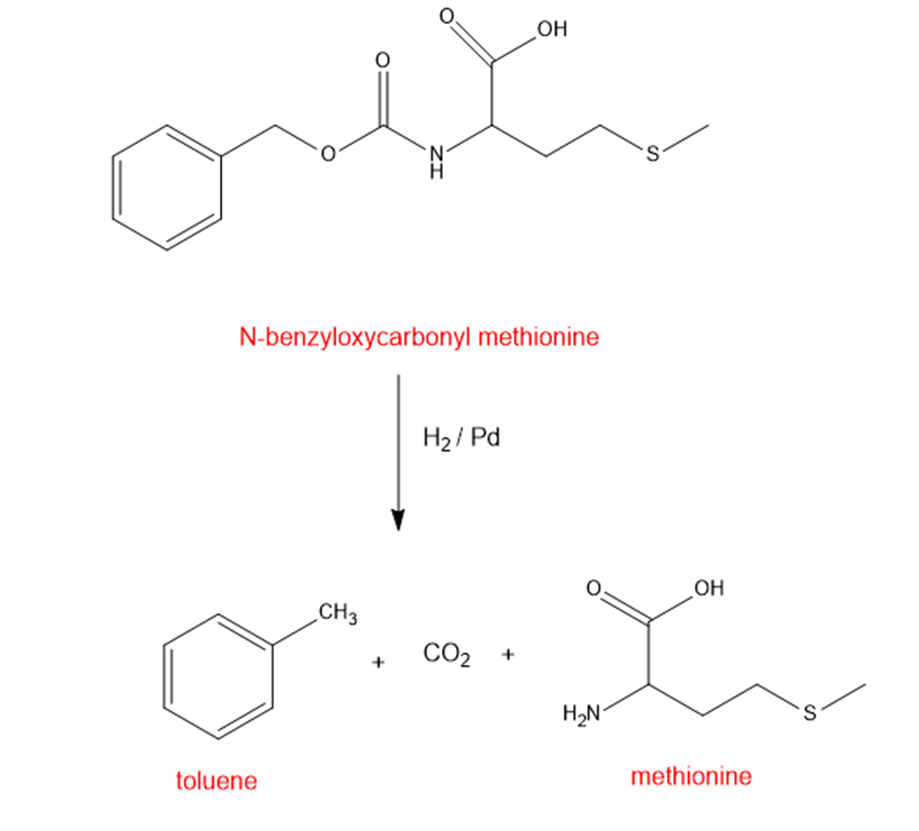
Hydrogenolysis of N-benzyloxy carbonyl methionine
Learning Materials
Features
Discover
Chapter 24: Q16P. (page 1273)
Give equations for the formation and hydrogenolysis of N-benzyloxy carbonyl methionine.
 Formation of N- benzyloxy carbonyl methionine
Formation of N- benzyloxy carbonyl methionine

Hydrogenolysis of N-benzyloxy carbonyl methionine
All the tools & learning materials you need for study success - in one app.
Get started for free
A molecular weight determination has shown that an unknown peptide is a pentapeptide, and an amino acid analysis shows that it contains the following residues: one Gly, two Ala, one Met, one Phe. Treatment of the original pentapeptide with carboxypeptidase gives alanine as the first free amino acid released. Sequential treatment of the pentapeptide with phenyl isothiocyanate followed by mild hydrolysis gives the following derivatives:
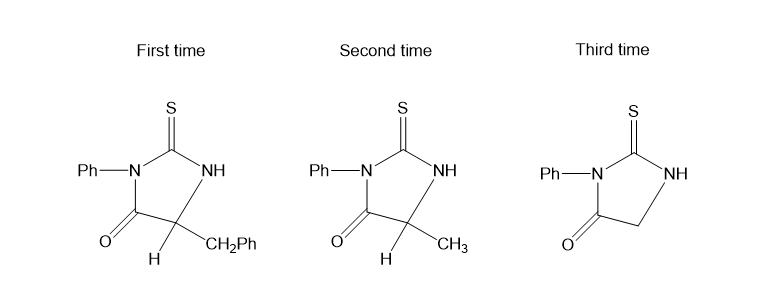
Propose a structure for the unknown pentapeptide.
Show how the following amino acids might be formed in the laboratory by reductive amination of the appropriate -ketoacid.
Phenylalanine (b) Cysteine (c) Serine (d) Alanine
The herbicide glyphosate(Roundup) kills plants by inhibiting an enzyme needed for synthesis of phenylalanine. Deprived of phenylalanine, the plant cannot make the proteins it needs, and it gradually weakens and dies. Although a small amount of glyphosate is deadly to a plant, its human toxicity is quite low. Suggest why this powerful herbicide has little effect on humans.
Draw the resonance forms of a protonated guanidino group and explain why arginine has such a strongly basic isoelectric point.
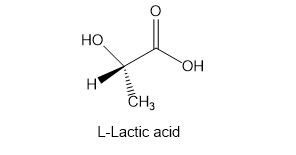
Suggest a method for the synthesis of the natural L enantiomer of alanine from the readily available L enantiomer of lactic acid.
What do you think about this solution?
We value your feedback to improve our textbook solutions.