Chapter 19: Q38P (page 1029)
Predict the products of the following reactions:
(a)
(b)
(c)
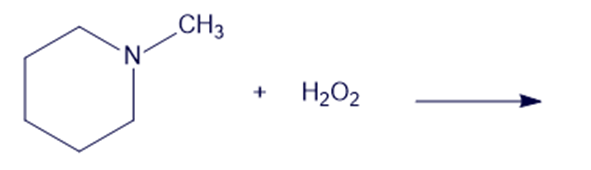
(d)
(e)
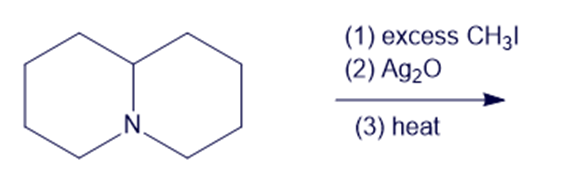
(f)
(g)
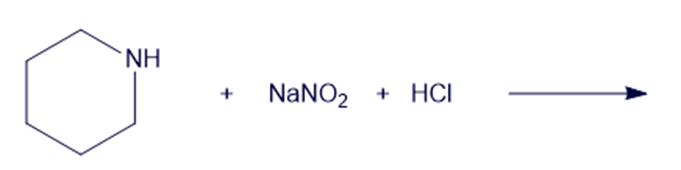
(h)
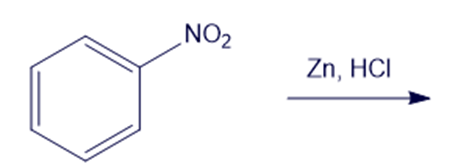
(i)
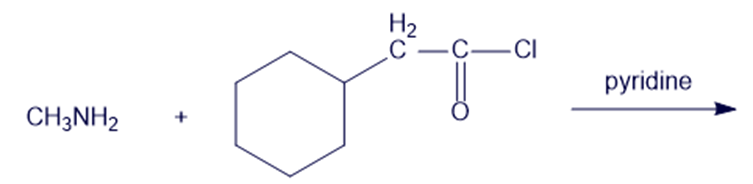
(j)
(k)
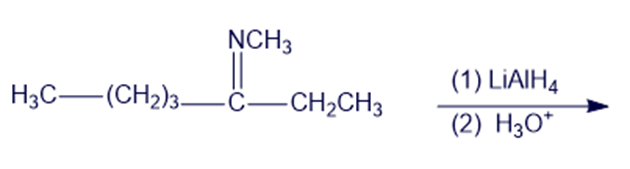
(l)
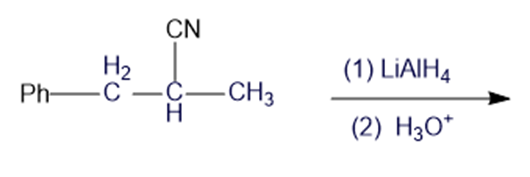
Short Answer
(a)
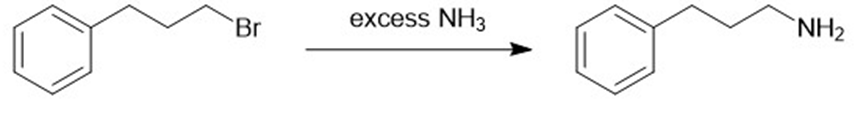
(b)

(c)
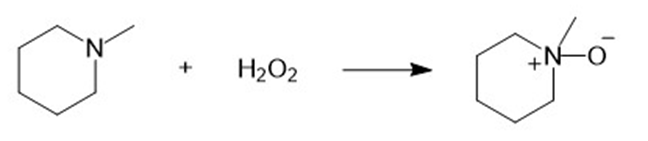
(d)
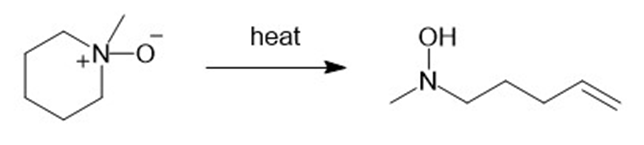
(e)
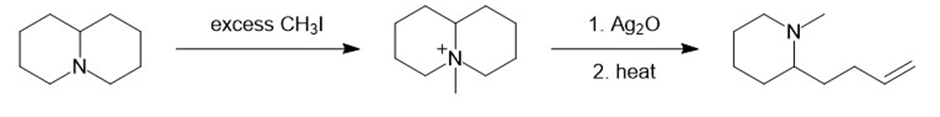
(f)
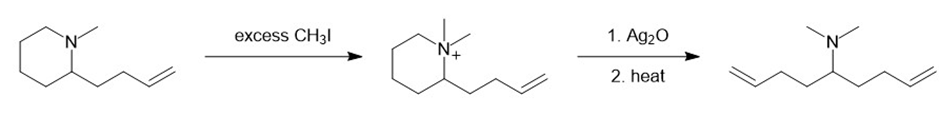
(g)

(h)
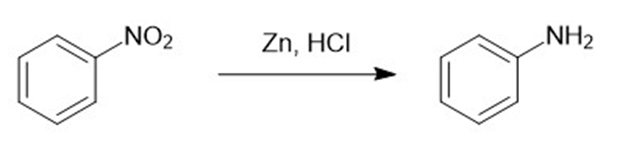
(i)
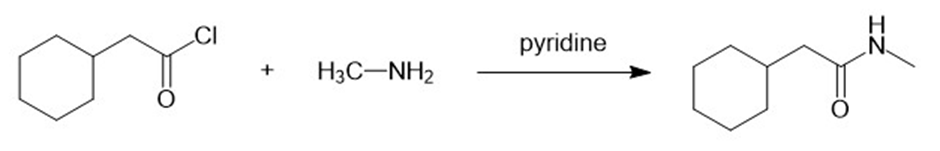
(j)
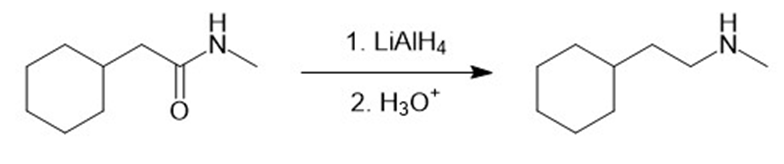
(k)
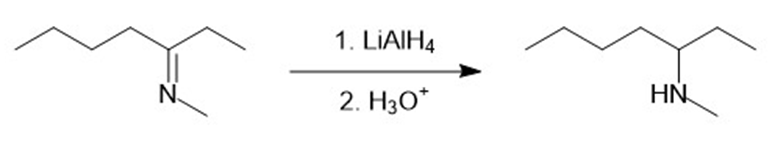
(l)
Learning Materials
Features
Discover
Chapter 19: Q38P (page 1029)
Predict the products of the following reactions:
(a)
(b)
(c)
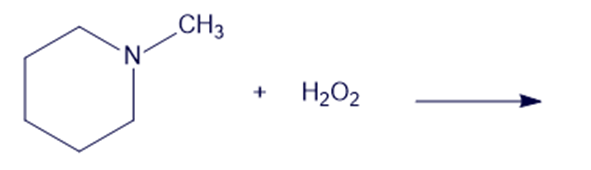
(d)
(e)

(f)
(g)

(h)

(i)

(j)
(k)

(l)

(a)

(b)

(c)
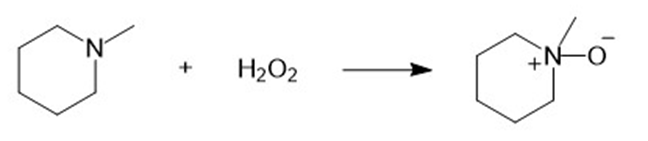
(d)

(e)

(f)

(g)

(h)

(i)

(j)

(k)

(l)

All the tools & learning materials you need for study success - in one app.
Get started for free
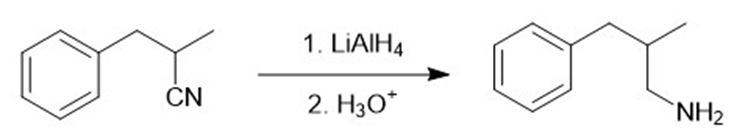
The two most general amine syntheses are the reductive amination of carbonyl compounds and the reduction of amides. Show how these techniques can be used to accomplish the following syntheses.
(a) benzoic acid to benzylamine
(b) benzaldehyde to benzylamine
(c) pyrrolidine to N-ethylpyrrolidine
(d) cyclohexanone to N- cyclohexylpyrrolidine
(e) HOOC-(CH2)3 -COOH to pentane-1,5-diamine cadaverine
(A true story.) A drug user responded to an ad placed by a DEA informant in a drug-culture magazine. He later flew from Colorado to Maryland, where he bought some 1-phenyl-2-propanone (P2P) from the informant. The police waited nearly a month for the suspect to synthesize something, then obtained a search warrant, and searched the residence. They found the unopened bottle of P2P; apparently, the suspect was not a good chemist and was unable to follow the instructions the informant gave him. They also found pipes and bongs with residues of marijuana and cocaine, plus a bottle of methylamine hydrochloride, some muriatic acid (dilute HCL), zinc strips, flasks, and other equipment.
(a) Assume you are consulting for the police. Show what synthesis the suspect was prepared to carry out, to provide probable cause for the charge of attempting to manufacture a controlled substance.
(b) Assume you are a member of the jury. Would you convict the defendant of attempting to manufacture a controlled substance?
Propose a mechanism for nitration of pyridine at the 4-position, and show why this orientation is not observed.
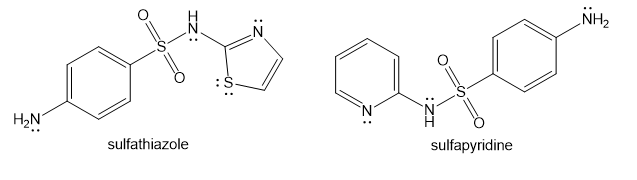
Show how you would use the same sulfonyl chloride as used in the sulfanilamide synthesis to make sulfathiazole and sulfapyridine.
Question. Propose a mechanism for the sulfonation of pyridine, and point out why sulfonation occurs at 3-position.
What do you think about this solution?
We value your feedback to improve our textbook solutions.