Chapter 19: Q19P (page 1003)
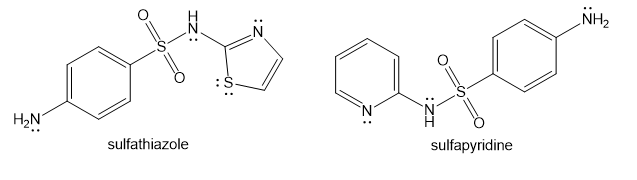
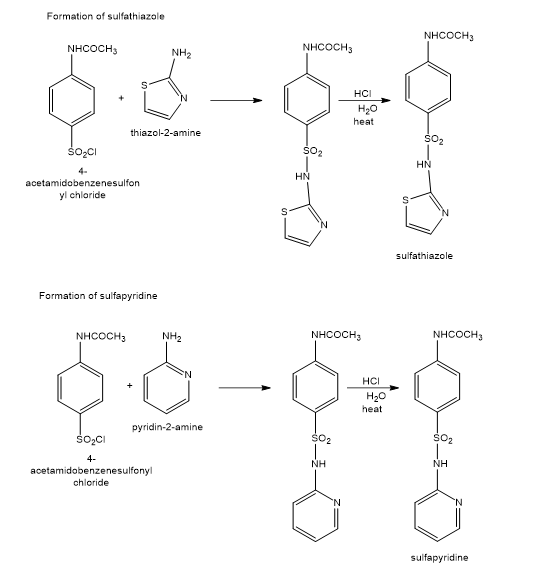
Show how you would use the same sulfonyl chloride as used in the sulfanilamide synthesis to make sulfathiazole and sulfapyridine.
Short Answer
Learning Materials
Features
Discover
Chapter 19: Q19P (page 1003)
Show how you would use the same sulfonyl chloride as used in the sulfanilamide synthesis to make sulfathiazole and sulfapyridine.


All the tools & learning materials you need for study success - in one app.
Get started for free
Determine which of the heterocyclic amines just shown are not aromatic. Give the reasons for your conclusions.
Basicity depends on availability of an electron pair to bond a proton. Correlate structural effects in these amines with their basicities.
(a) Explain this order:
Give the products expected from the following reactions:
(a) acetyl chloride + ethylamine
(b) benzoyl chloride +dimethylamine
(c) hexanoyl chloride + piperidine
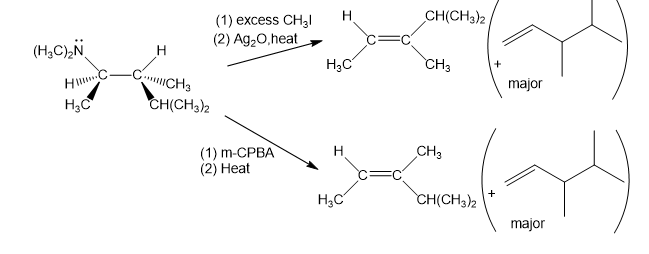
When the (R,R) isomer of the amine shown is treated with an excess of methyl iodide, then silver oxide, then heated, the major product is the Hofman product.
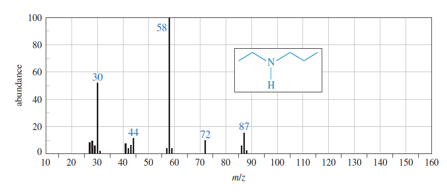
(a) Show how fragmentation occurs to give the base peak at m/z 58 in the mass spectrum of ethyl propyl amine (N-ethylpropan-1-amine), shown below.
(b) Show how a similar cleavage in the ethyl group gives an ion of m/z72.
(c) Explain why the peak at m/z 72 is much weaker than the one at m/z 58.
What do you think about this solution?
We value your feedback to improve our textbook solutions.