Chapter 19: Q17P (page 1001)
Give the products expected from the following reactions:
(a) acetyl chloride + ethylamine
(b) benzoyl chloride +dimethylamine
(c) hexanoyl chloride + piperidine
Short Answer
(a)
(b)
(c)
Learning Materials
Features
Discover
Chapter 19: Q17P (page 1001)
Give the products expected from the following reactions:
(a) acetyl chloride + ethylamine
(b) benzoyl chloride +dimethylamine
(c) hexanoyl chloride + piperidine
(a)
(b)
(c)
All the tools & learning materials you need for study success - in one app.
Get started for free
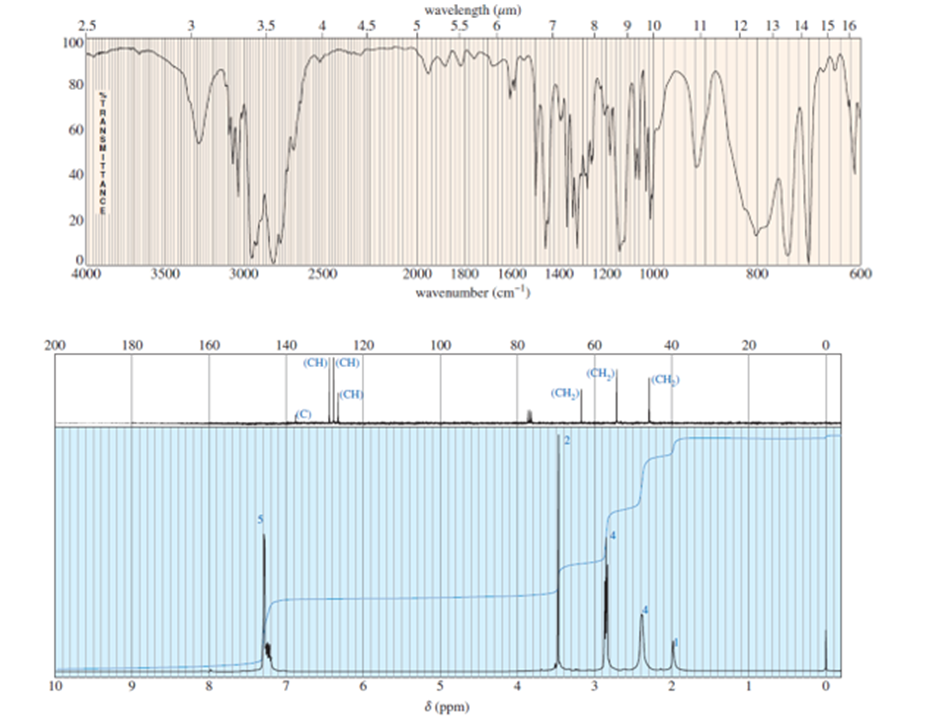
A compound of formula C11H16N2gives the IR, 1HNMR, and 13CNMR spectra shown. The proton NMR peak atδ 2.0disappears on shaking with D2O. Propose a structure for this compound, and show how your structure accounts for the observed absorptions.
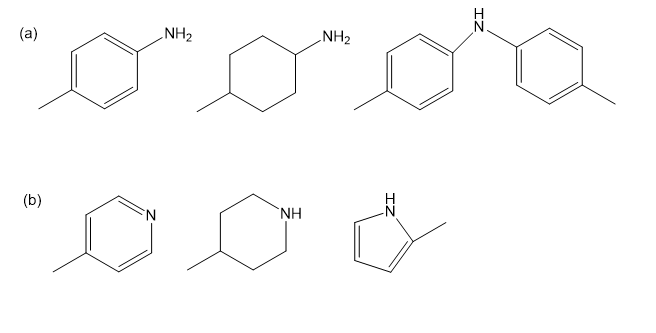
Rank the amines in each set in order of increasing basicity.
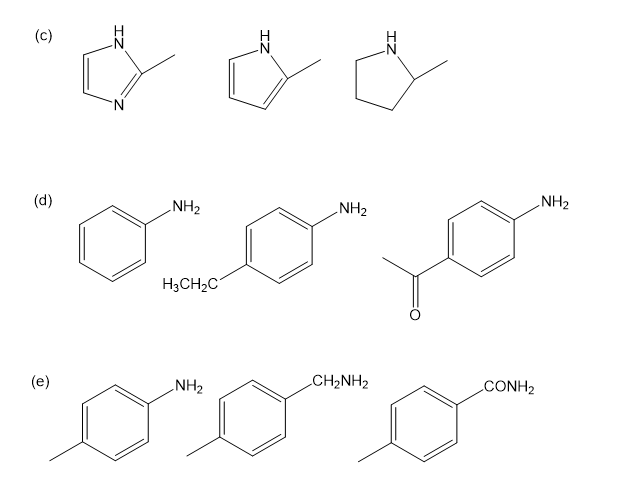
Predict the products from the reactions of the following amines with sodium nitrite in dilute.
In Section 19-10B, we saw that pyridine undergoes electrophilic aromatic substitution reluctantly, requiring strong conditions and giving disappointing yields. In contrast, pyridine N-oxide undergoes EAS under moderate conditions, giving good yields of substitution on C2 and C4. Explain this surprising difference.
When the (R,R) isomer of the amine shown is treated with an excess of methyl iodide, then silver oxide, then heated, the major product is the Hofman product.
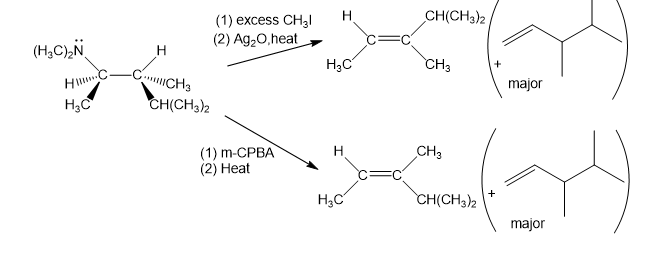
What do you think about this solution?
We value your feedback to improve our textbook solutions.