Chapter 19: Q39P (page 1030)
Predict the products of the following reactions:
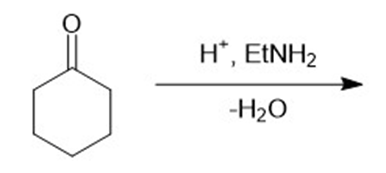
h.
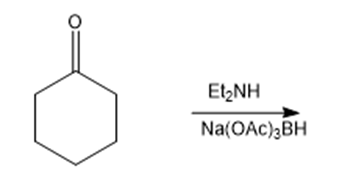
i.
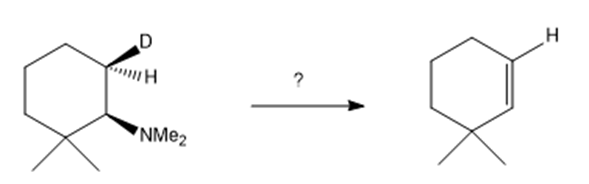
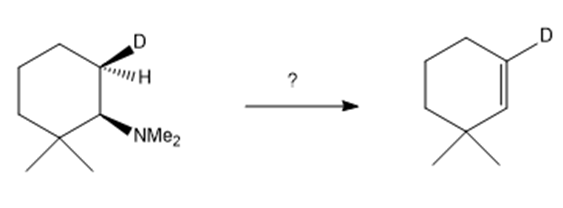
j.
Short Answer
a.
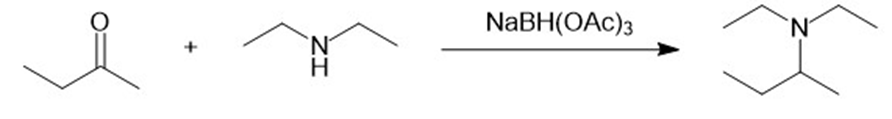
b.
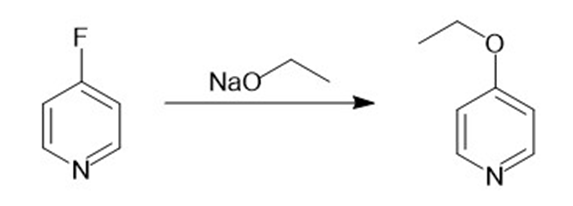
c.
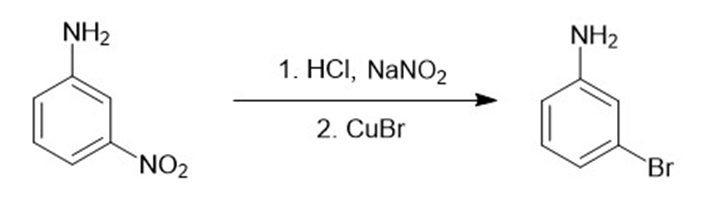
d.
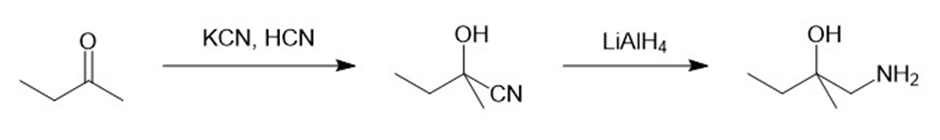
e.
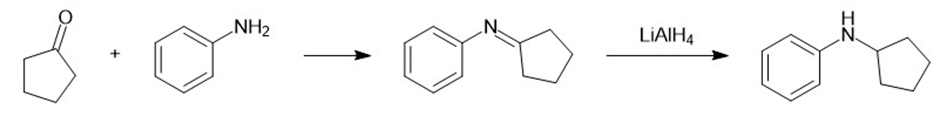
f.
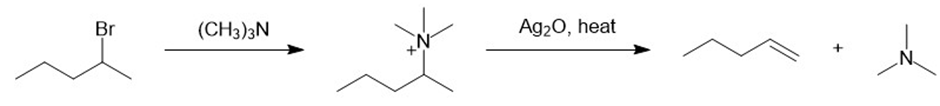
g.
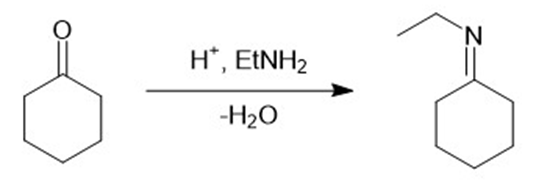
h.
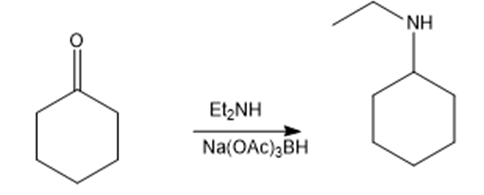
i.
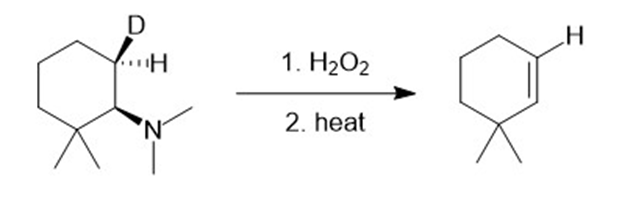
j.
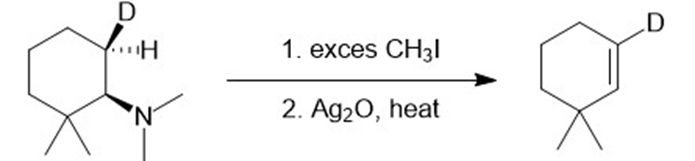
Learning Materials
Features
Discover
Chapter 19: Q39P (page 1030)
Predict the products of the following reactions:

h.

i.

j.

a.

b.

c.

d.

e.

f.

g.

h.

i.

j.
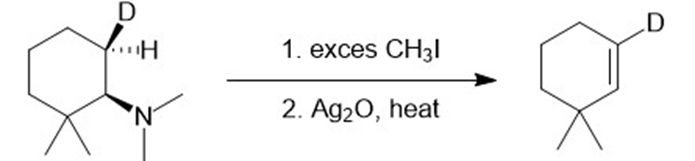
All the tools & learning materials you need for study success - in one app.
Get started for free
The two most general amine syntheses are the reductive amination of carbonyl compounds and the reduction of amides. Show how these techniques can be used to accomplish the following syntheses.
(a) benzoic acid to benzylamine
(b) benzaldehyde to benzylamine
(c) pyrrolidine to N-ethylpyrrolidine
(d) cyclohexanone to N- cyclohexylpyrrolidine
(e) HOOC-(CH2)3 -COOH to pentane-1,5-diamine cadaverine
Addition of one equivalent of ammonia to 1-bromoheptane gives a mixture of heptan-1-amine, some dialkylamine, some trialkylamine, and even some tetraalkylammonium bromide.
Reductive amination of aldehydes and ketones is a versatile method for attaching alkyl groups to amines, but the alkyl group is restricted to a 1 0or 20carbon by this method. Prof. Phil Baran of Scripps Research Institute has reported (Science, 2015, 348(6237), 886-891) a novel way to reduce an aromatic nitro group and add the resulting amine to an alkene so that the aromatic amine is bonded to a carbon- all in a continuous sequence of reactions. For example:
Predict the products using these starting materials, all of which are reported in this paper.
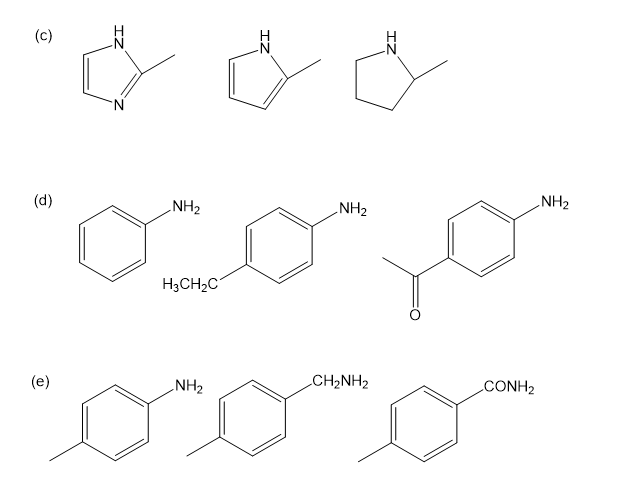
Rank the amines in each set in order of increasing basicity.
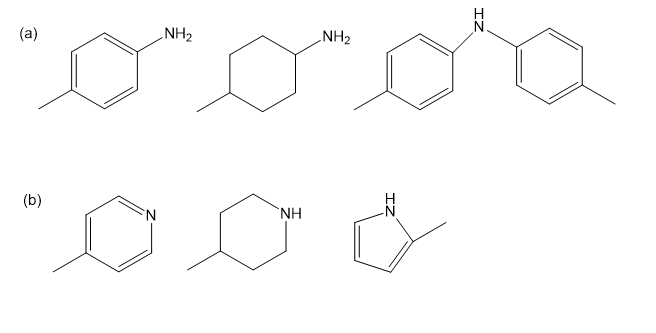
a) Guanidine (shown) is about as strong base as hydroxide ion. Explain why guanidine is a much stronger base than most other amines.
(b) Show why p-nitroaniline is a much weaker base (3 pKb units weaker) than aniline.
(c) Explain why N,N-2,6-tetramethylaniline (shown) is a much stronger base than N,N-dimethylaniline.
What do you think about this solution?
We value your feedback to improve our textbook solutions.