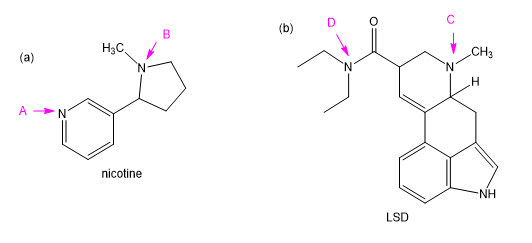
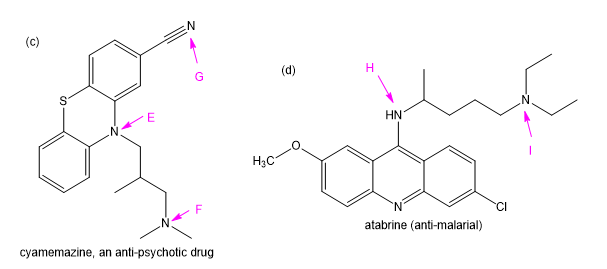
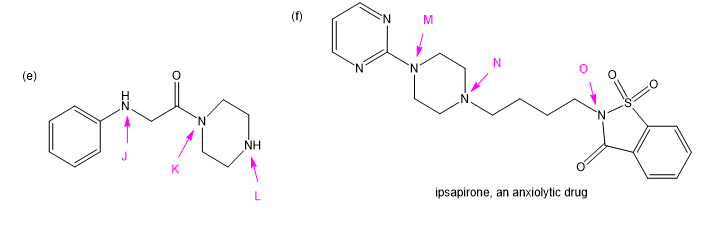
Chapter 19: Q11P (page 997)
Propose a mechanism for nitration of pyridine at the 4-position, and show why this orientation is not observed.
Short Answer
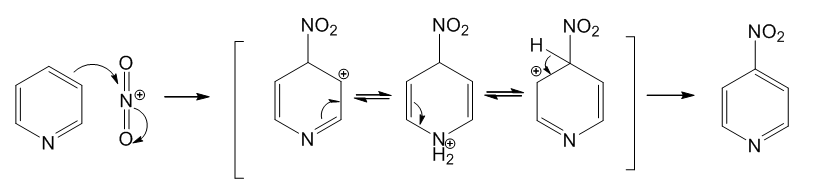
This orientation is not observed because electrophilic substitution reaction at 4-position is not stable. The intermediate formed by electrophilic addition at 4-position is less stable than that of 3-position.