Chapter 27: Pericyclic Reactions
Q 38.
Question: What type of cycloaddition occurs in Reaction [1]? Draw the product of a similar process in Reaction [2]. Would you predict that these reactions occur under thermal or photochemical conditions?
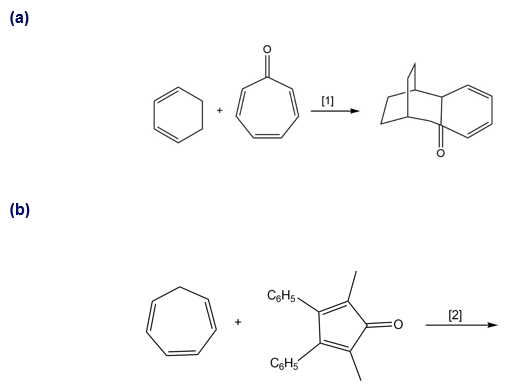
Q 39.
Question: Draw the product of each Diels–Alder reaction and indicate the stereochemistry at all stereogenic centers
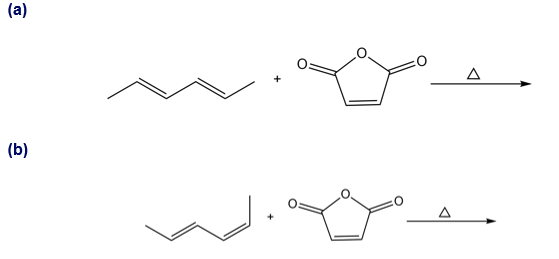
Q 4.
Question: (a) How many π molecular orbitals are present in deca-1,3,5,7,9-pentaene ? (b) How many are bonding MOs and how many are antibonding MOs ? (c) How many nodes are present in ? (d) How many nodes are present in ?
Q 40.
Question: What cycloaddition products are formed in each reaction? Indicate the stereochemistry of each product.
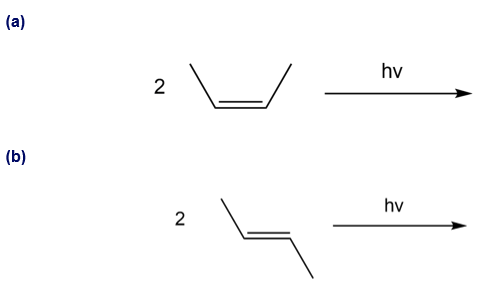
Q 41.
Question:What starting materials are needed to synthesize each compound by a thermal [4 + 2] cycloaddition?
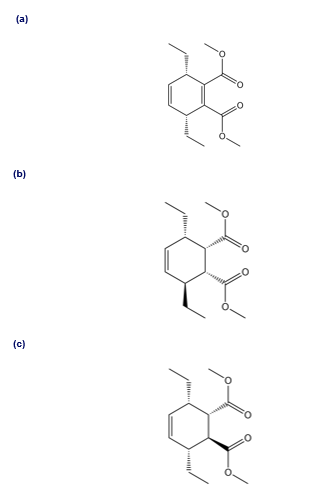
Q 42.
Question: Explain why heating buta-1,3-diene forms 4-vinylcyclohexene but not cycloocta-1,5-diene.
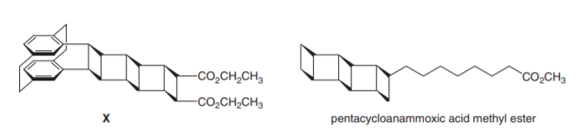
Q 43.
Question: How can X be prepared from a constitutional isomer by a series of cycloaddition reactions? Interest in molecules that contain several cyclobutane rings fused together has been fueled by the discovery of pentacycloanammoxic acid methyl ester, a lipid isolated from the membrane of organelles in the bacterium Candidatus Brocadia anammoxidans. The role of this unusual natural product is as yet unknown.
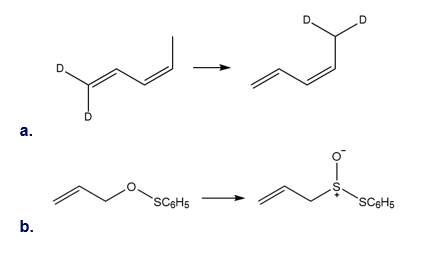
Q 44.
Question: What type of sigmatropic rearrangement is illustrated in each reaction?
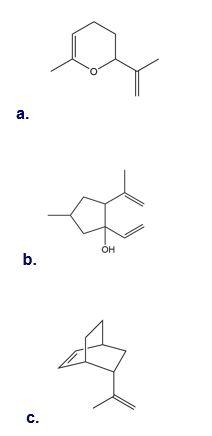
Q 45.
Question: Draw the product of the [3,3] sigmatropic rearrangement of each compound.
Q 46.
Question: A solution of 5-methylcyclopenta-1,3-diene rearranges at room temperature to a mixture containing 1-methyl-, 2-methyl-, and 5-methylcyclopenta-1,3-diene. (a) Show how both isomeric products are formed from the starting material by a sigmatropic rearrangement involving a C–H bond. (b) Explain why 2-methylcyclopenta-1,3-diene is not formed directly from 5-methylcyclopenta-1,3-diene by a [1,3] rearrangement.
