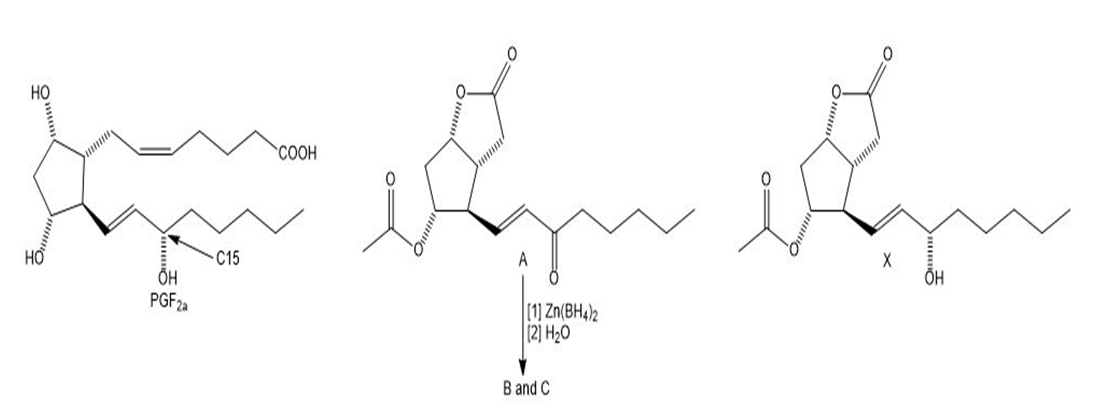
Chapter 31: Question 31.9 (page 1242)
Explain why regularly ingesting a large excess of a fat-soluble vitamin can lead to severe health problems, whereas ingesting a large excess of a water-soluble vitamin often causes no major health problems.
Short Answer
Answer
Fat-soluble vitamins are not excreted from the body with water. Hence, ingesting a fat-soluble vitamin can cause severe health problems.
Water-soluble vitamins are easily excreted out from the body with water. Hence, ingesting a water-soluble vitamin does not cause health problems.