Chapter 31: Q31P (page 1259)
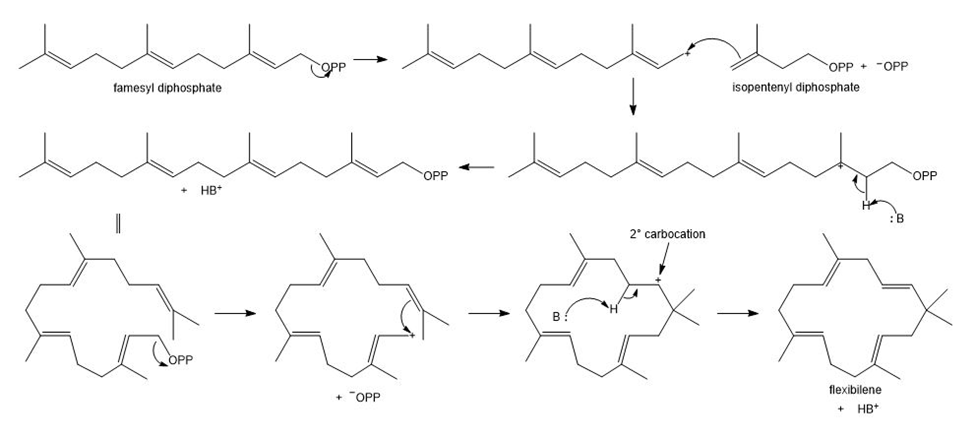
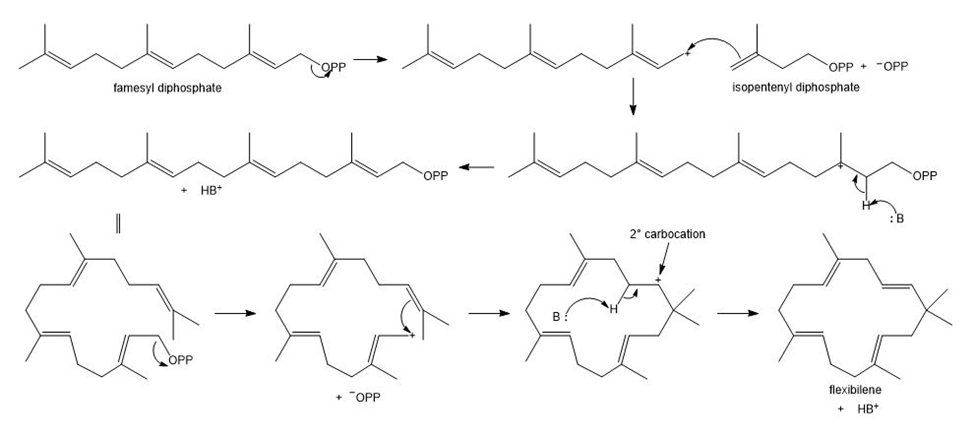
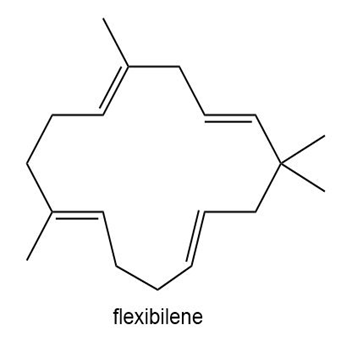
Flexibilene is a terpene isolated from Sinulariaflexibilis, a soft coral found in the Indian Ocean. Draw a stepwise mechanism for the formation of flexibilene from farnesyl diphosphate and isopentenyl diphosphate. What is unusual about the cyclization that forms the 15-membered ring of flexibilene?
Short Answer
The unusual feature of the cyclization is the generation of carbocation instead of
carbocation.