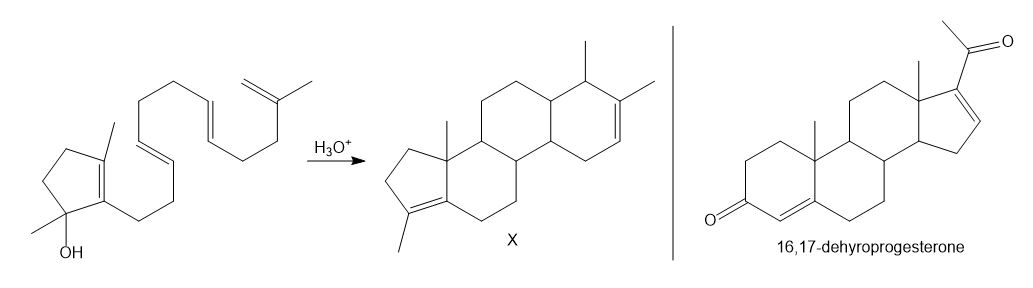
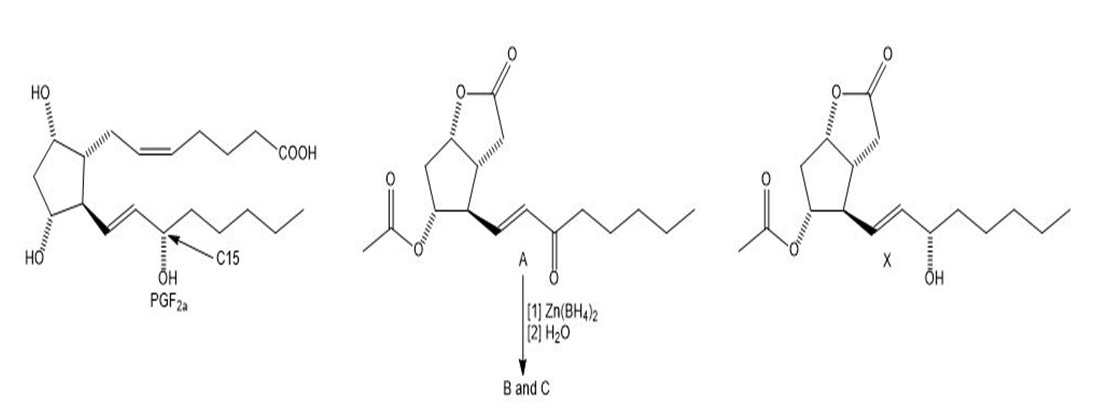
Chapter 31: Question 31.5 (page 1237)
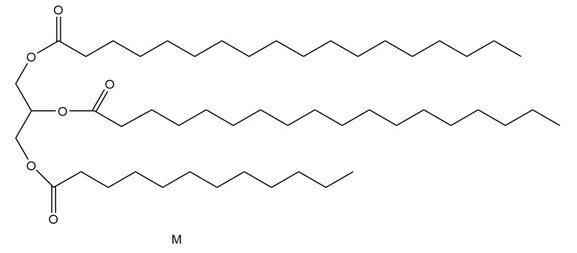
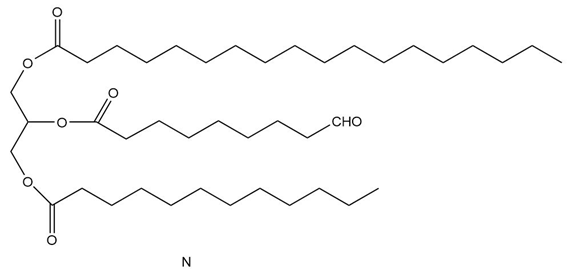
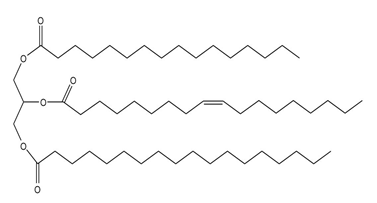
Unlike many fats and oils, the cocoa butter used to make chocolate is remarkably uniform in composition. All triacylglycerols contain oleic acid esterified to the OH group of glycerol, and either palmitic acid or stearic acid esterified to the OH groups. Draw the structures of two possible triacylglycerols that compose cocoa butter.
Short Answer
Answer
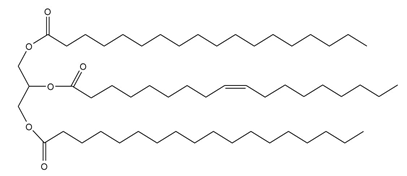
 Structure of the first possible triacylglycerol that composes cocoa butter
Structure of the first possible triacylglycerol that composes cocoa butter Structure of the second possible triacylglycerol that composes cocoa butter
Structure of the second possible triacylglycerol that composes cocoa butter