Chapter 31: Q41P (page 1260)
Farnesyl diphosphate is cyclized to sesquiterpene A, which is then converted to the bicyclic product epi-aristolochene. Write a stepwise mechanism for both reactions.
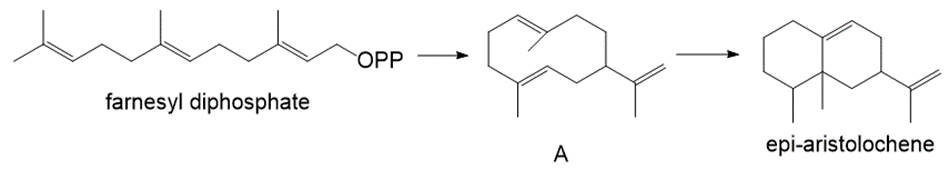
Short Answer
Answer
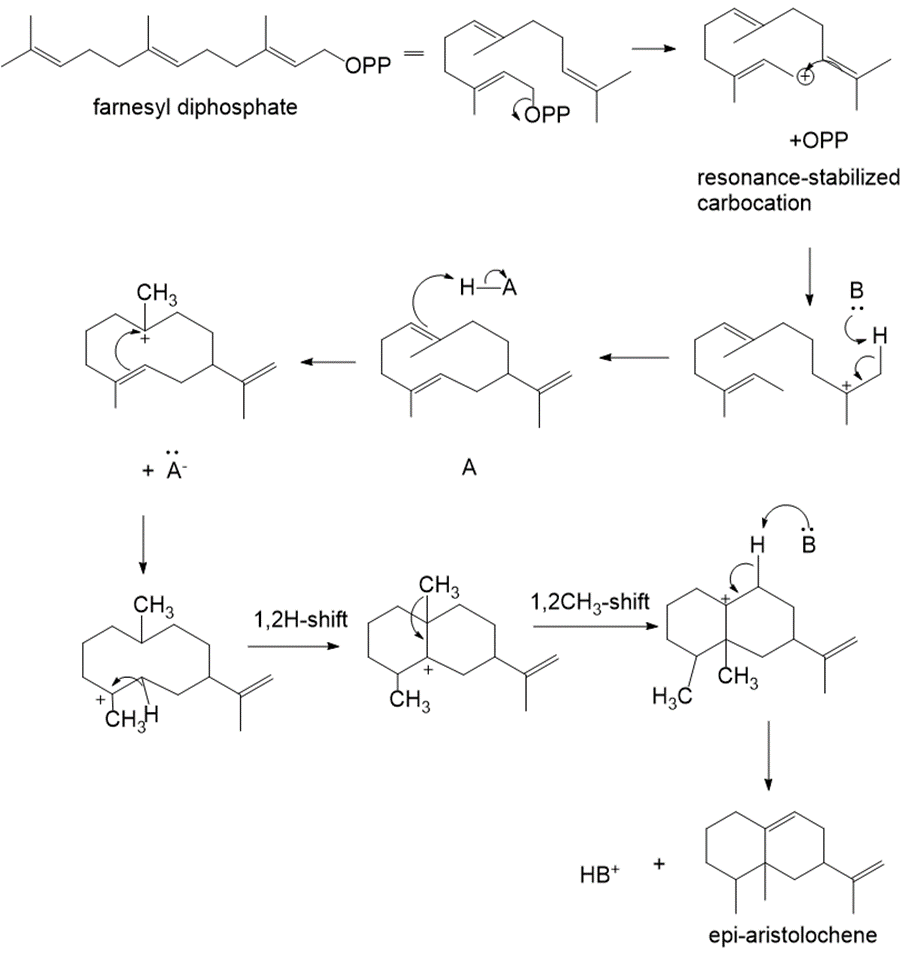
The mechanism of the reaction
Learning Materials
Features
Discover
Chapter 31: Q41P (page 1260)
Farnesyl diphosphate is cyclized to sesquiterpene A, which is then converted to the bicyclic product epi-aristolochene. Write a stepwise mechanism for both reactions.

Answer

The mechanism of the reaction
All the tools & learning materials you need for study success - in one app.
Get started for free
How would you expect the melting point of eicosapentaenoic acid to compare with the melting points of the fatty acids listed in Table 31.2?
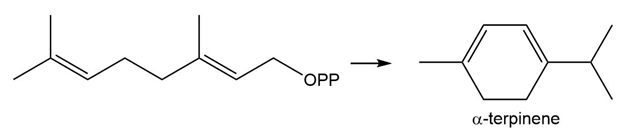
Draw a stepwise mechanism for the conversion of geranyldiphosphateto -terpinene .
Phosphoacylglycerols should remind you of soaps (Section 3.6). In what ways are these compounds similar?
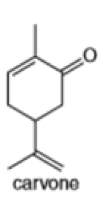
Locate the isoprene units in each compound.
(a) (b)
(b)
(c)
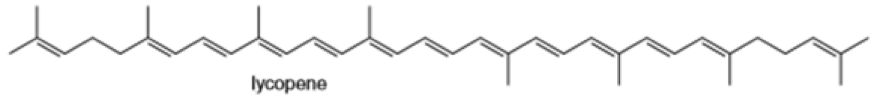
(d)
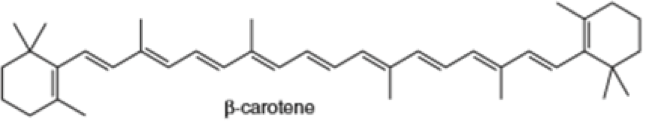
(e) (f)
(f)
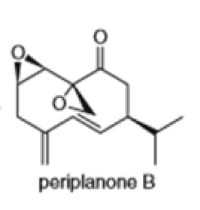
(g)
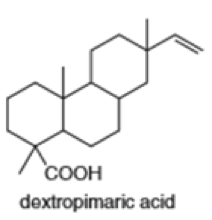
(h)
Explain why regularly ingesting a large excess of a fat-soluble vitamin can lead to severe health problems, whereas ingesting a large excess of a water-soluble vitamin often causes no major health problems.
What do you think about this solution?
We value your feedback to improve our textbook solutions.