Chapter 31: Q26P. (page 1258)
Locate the isoprene units in each compound.
(a) (b)
(b)
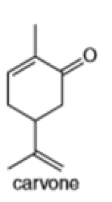
(c)
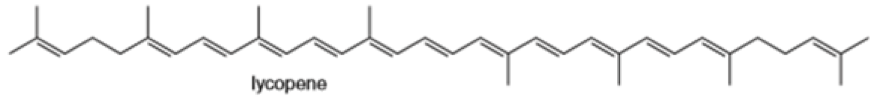
(d)
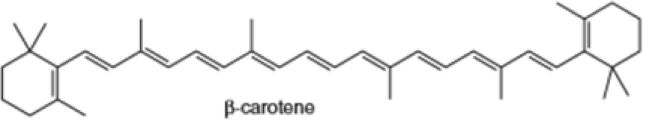
(e) (f)
(f)
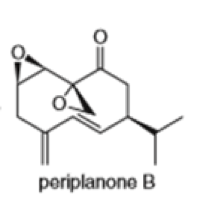
(g)
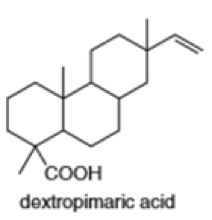
(h)
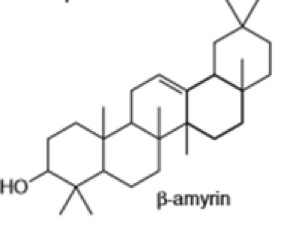
Short Answer
(a)
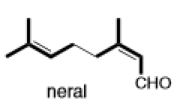
(b)
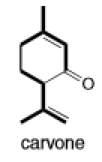
(c)
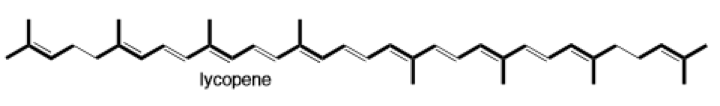
(d)
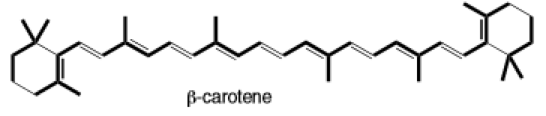
(e)
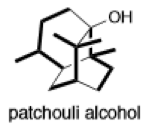
(f)
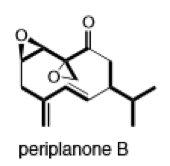
(g)
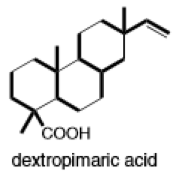
(h)
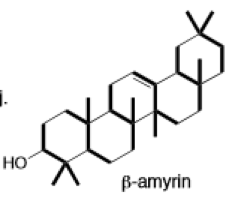
Learning Materials
Features
Discover
Chapter 31: Q26P. (page 1258)
Locate the isoprene units in each compound.
(a) (b)
(b)

(c)

(d)

(e) (f)
(f)

(g)

(h)

(a)

(b)

(c)

(d)

(e)

(f)

(g)

(h)

All the tools & learning materials you need for study success - in one app.
Get started for free
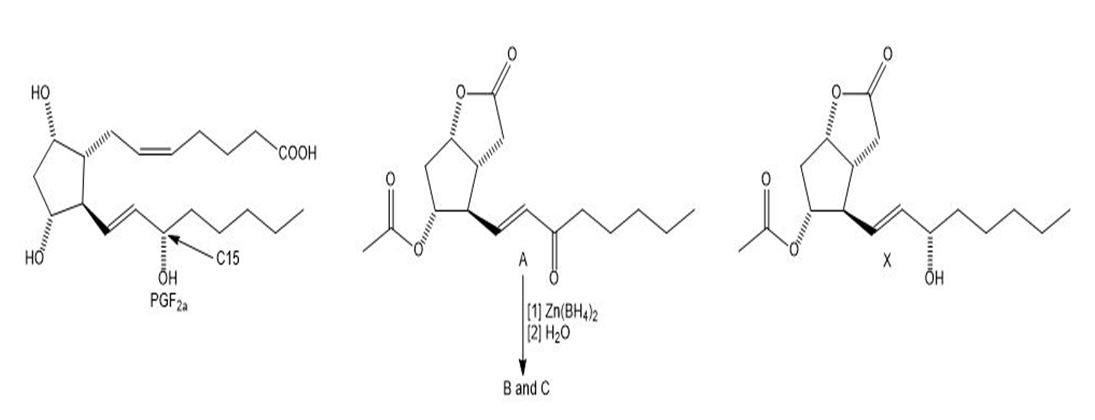
A difficult problem in the synthesis of PGFis the introduction of the OH group at C15 in the desired configuration.
A well known synthesis of PGFinvolves reaction of A with (a metal hydride reagent similar in reactivity to, to form two isomeric products, B and C. Draw their structures and indicate their stereochemical relationship.
Suggest a reagent to convert A to the single stereoisomer X.
Unlike many fats and oils, the cocoa butter used to make chocolate is remarkably uniform in composition. All triacylglycerols contain oleic acid esterified to the OH group of glycerol, and either palmitic acid or stearic acid esterified to the OH groups. Draw the structures of two possible triacylglycerols that compose cocoa butter.
Draw the enantiomer and any two diastereomers of cholesterol. Does the OH group of cholesterol occupy an axial or equatorial position?
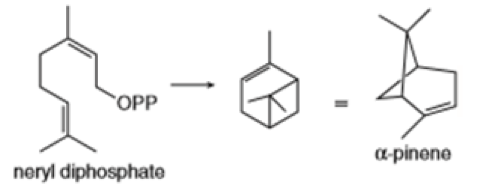
Draw a stepwise mechanism for the conversion of neryl diphosphate to is a component of pine oil and rosemary oil.
Explain why regularly ingesting a large excess of a fat-soluble vitamin can lead to severe health problems, whereas ingesting a large excess of a water-soluble vitamin often causes no major health problems.
What do you think about this solution?
We value your feedback to improve our textbook solutions.