Chapter 31: Q18P. (page 1256)
Convert each ball-and-stick model to a skeletal structure that clearly shows the stereochemistry at the ring fusion of these decalin derivatives.
A.
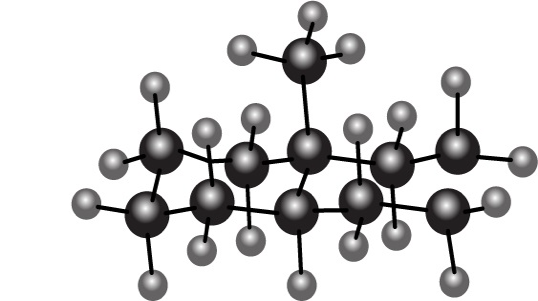
B.
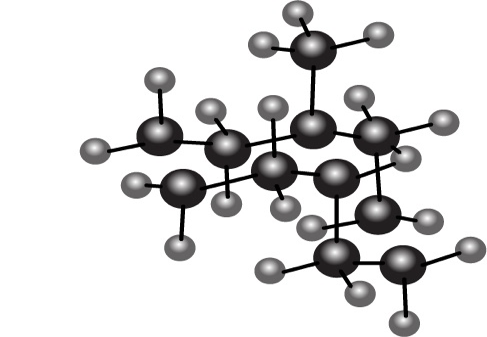
Short Answer
A.
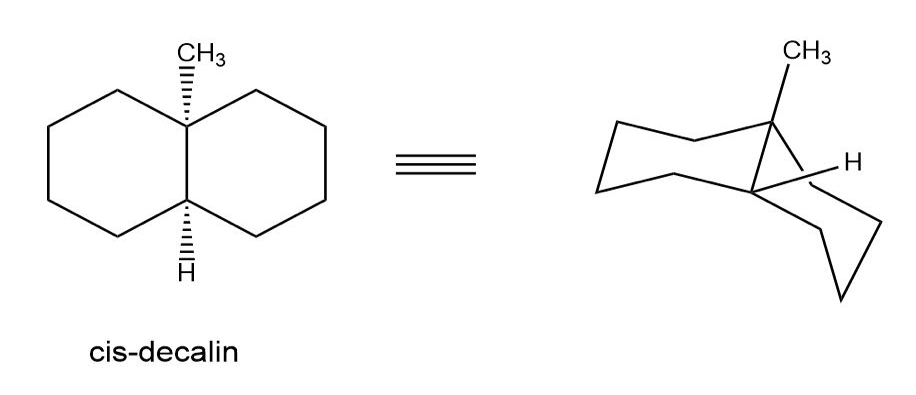
B.

Learning Materials
Features
Discover
Chapter 31: Q18P. (page 1256)
Convert each ball-and-stick model to a skeletal structure that clearly shows the stereochemistry at the ring fusion of these decalin derivatives.
A.

B.

A.

B.

All the tools & learning materials you need for study success - in one app.
Get started for free
Convert the ball-and-stick model of androsterone to (a) a skeletal structure using wedges and dashed wedges around all stereogenic centers; and (b) a three-dimensional representation using chair cyclohexane rings.
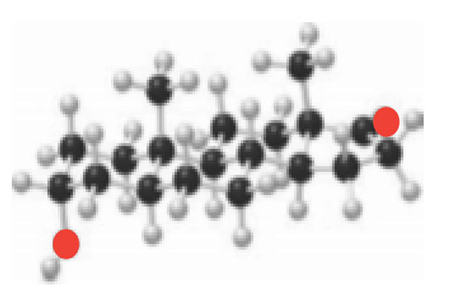
androsterone
The main fatty acid component of the triacylglycerols in coconut oil is lauric acid, . Explain why coconut oil is a liquid at room temperature even though it contains a large fraction of this saturated fatty acid.
Draw three-dimensional structures for each alcohol. Label the OH groups as occupying axial or equatorial positions.
a.
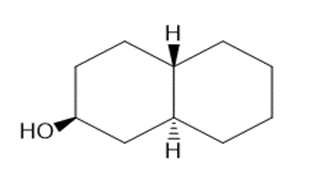
b.
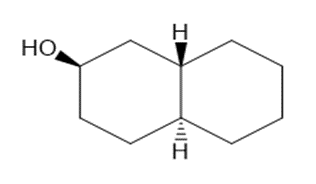
c.
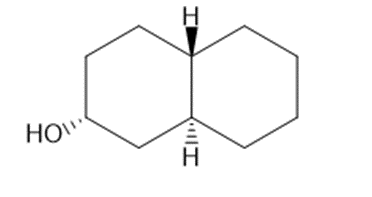
d.
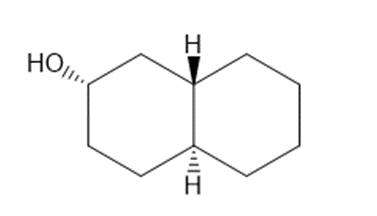
(a) Draw a skeletal structure of the anabolic steroid 4-androstene-3, 17-dione, also called “andro,” from the following description. Andro contains the tetracyclic steroid with carbonyl groups at C3 and C17, a double bond between C4 and C5, and methyl groups bonded to C10 and C13.
(b) Add wedges and dashed wedges for all stereogenic centers with the following information: the configuration at C10 is R, the configuration at C13 is S, and all substituents at ring fusions are trans to each other.
Question: A difficult problem in the synthesis of is the introduction of the OH group at C15 in the desired configuration.
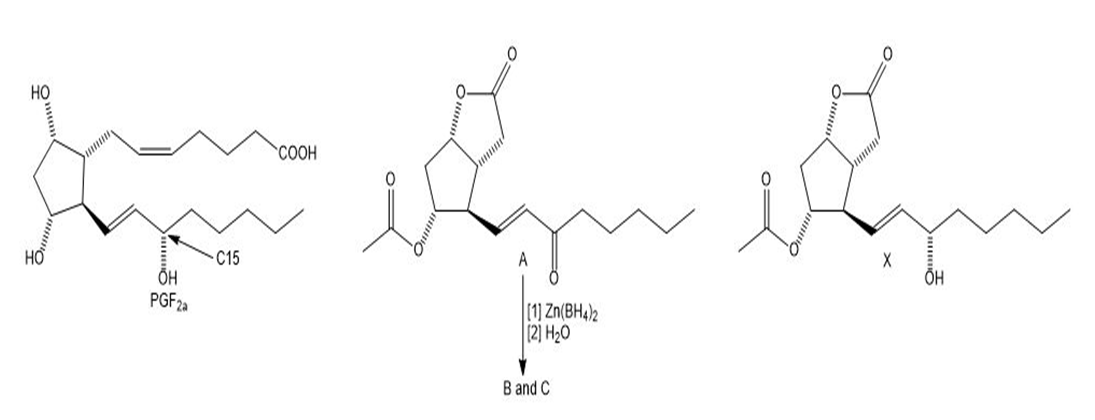
What do you think about this solution?
We value your feedback to improve our textbook solutions.