Chapter 31: Q33P. (page 1259)
Draw three-dimensional structures for each alcohol. Label the OH groups as occupying axial or equatorial positions.
a.
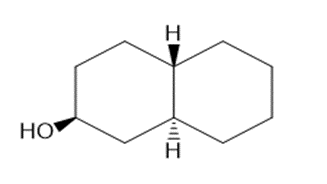
b.
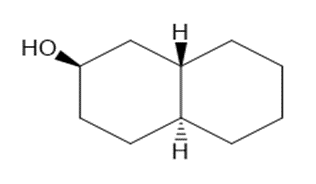
c.
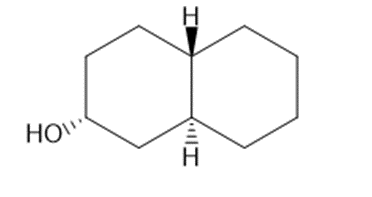
d.
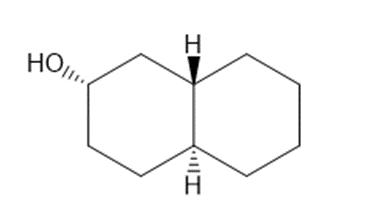
Short Answer
Answer
a.
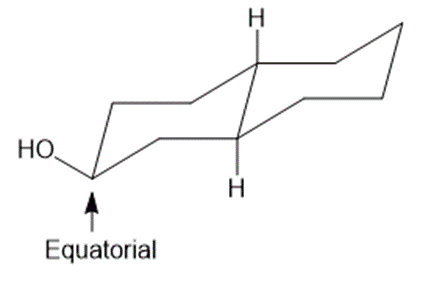
b.
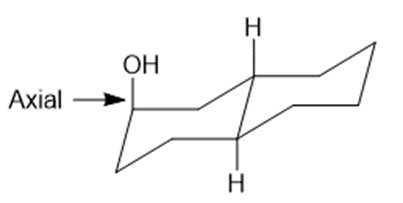
c.
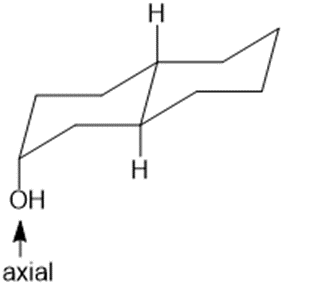
d.
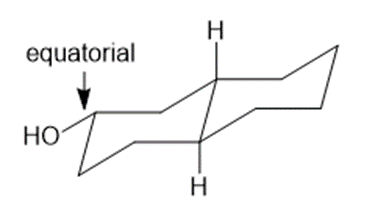
Learning Materials
Features
Discover
Chapter 31: Q33P. (page 1259)
Draw three-dimensional structures for each alcohol. Label the OH groups as occupying axial or equatorial positions.
a.

b.

c.

d.

Answer
a.

b.

c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
Phosphoacylglycerols should remind you of soaps (Section 3.6). In what ways are these compounds similar?
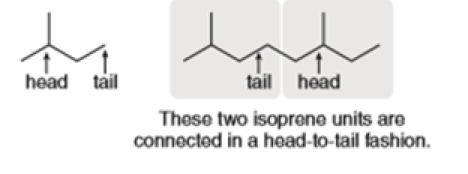
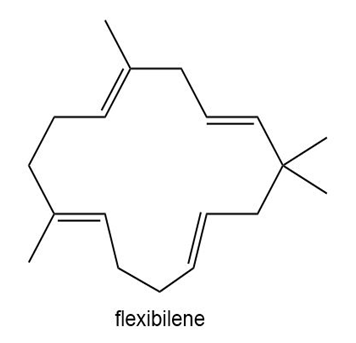
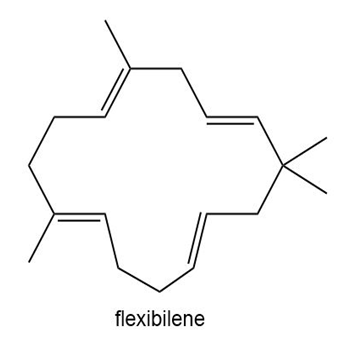
Question: Flexibilene is a terpene isolated from Sinulariaflexibilis, a soft coral found in the Indian Ocean. Draw a stepwise mechanism for the formation of flexibilene from farnesyl diphosphate and isopentenyl diphosphate. What is unusual about the cyclization that forms the 15-membered ring of flexibilene?
An isoprene unit can be thought of as having a head and a tail. The “head” of the isoprene unit is located at the end of the chain nearest the branch point, and the “tail” is located at the end of the carbon chain farthest from the branch point. Most isoprene units are connected together in a “head-to-tail” fashion, as illustrated. For both lycopene (Problem 31.26), and squalene (Figure 31.9), decide which isoprene units are connected in a head-to-tail fashion and which are not.
Draw all possible constitutional isomers of a triacylglycerol formed from one mole each of palmitic, oleic, and linoleic acids. Locate the tetrahedral stereogenic centers in each constitutional isomer.
What do you think about this solution?
We value your feedback to improve our textbook solutions.