Chapter 31: Q17P (page 1256)
Locate the isoprene units in each compound.
a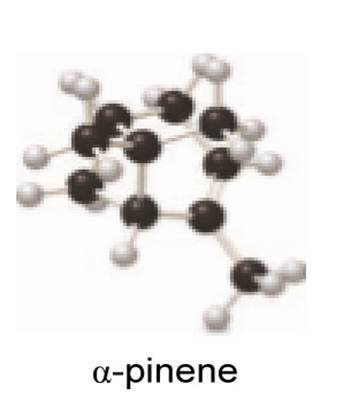
b.
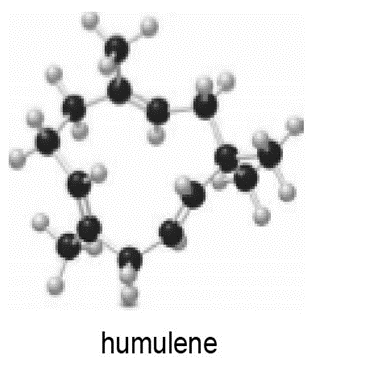
Short Answer
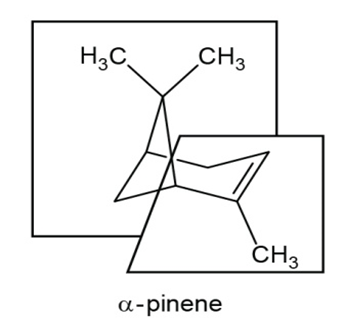
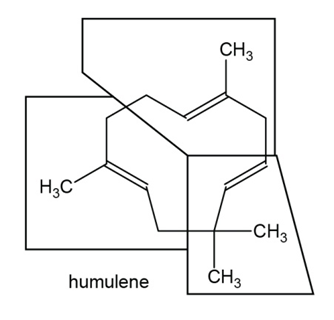
Answer
a.
b.
Learning Materials
Features
Discover
Chapter 31: Q17P (page 1256)
Locate the isoprene units in each compound.
a
b.

Answer
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
One component of lanolin, the wax that coats sheep’s wool, is derived from cholesterol and stearic acid. Draw its structure, including the correct stereochemistry of all stereogenic centers.
Draw a stepwise mechanism for the following reaction.
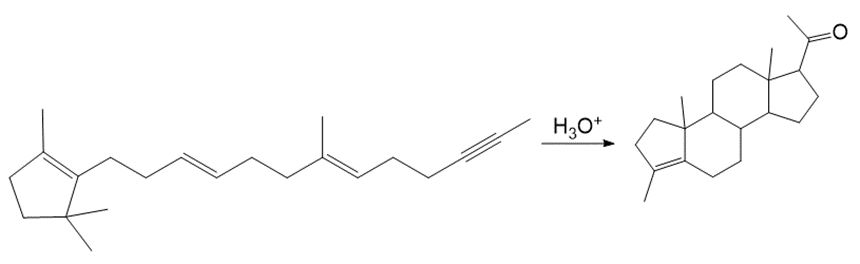
Draw a stepwise mechanism for the conversion of neryl diphosphate to is a component of pine oil and rosemary oil.
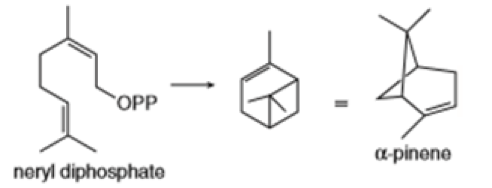
The biosynthesis of lanosterol from squalene has intrigued chemists since its discovery. It is now possible, for example, to synthesize polycyclic compounds from acyclic or monocyclic precursors by reactions that form several C-C bonds in a single reaction mixture.
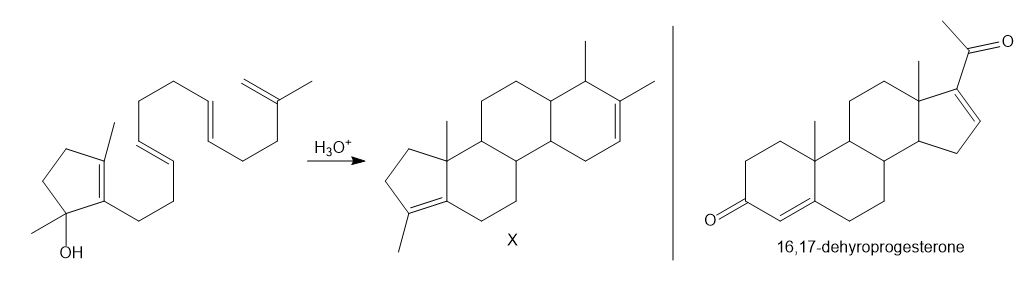
What is the structure of an optically inactive triacylglycerol that yields two moles of oleic acid and one mole of palmitic acid when hydrolyzed in aqueous acid?
What do you think about this solution?
We value your feedback to improve our textbook solutions.