Chapter 28: Q50P (page 1148)
Draw the products formed when D-altrose is treated with each reagent.

Short Answer
Answer
(a)
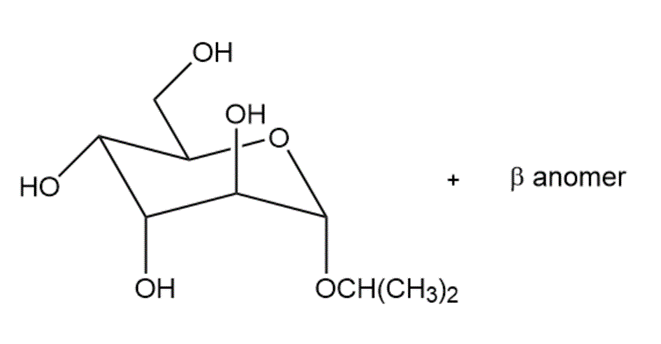
(b)
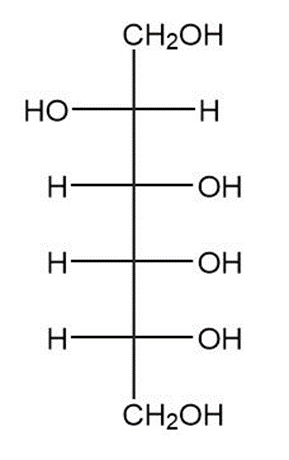
(c)
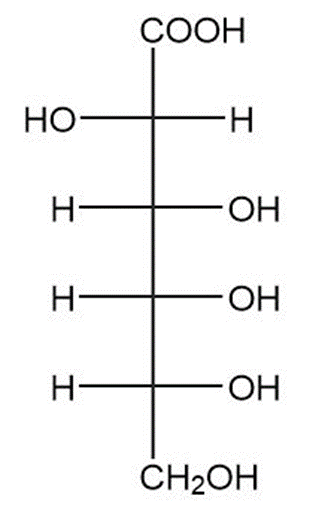
(d)
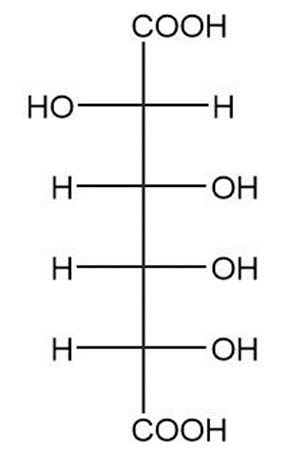
(e)
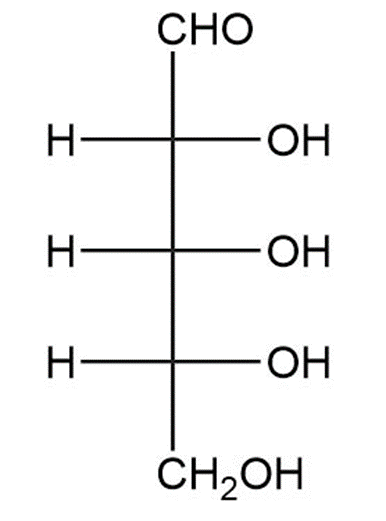
(f)
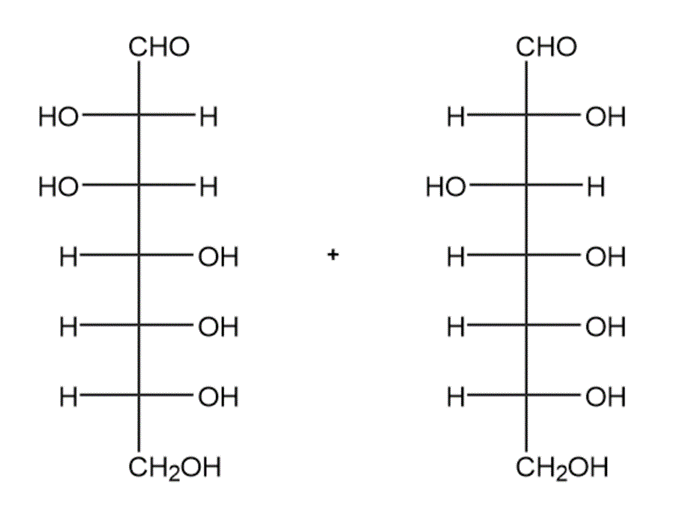
(g)
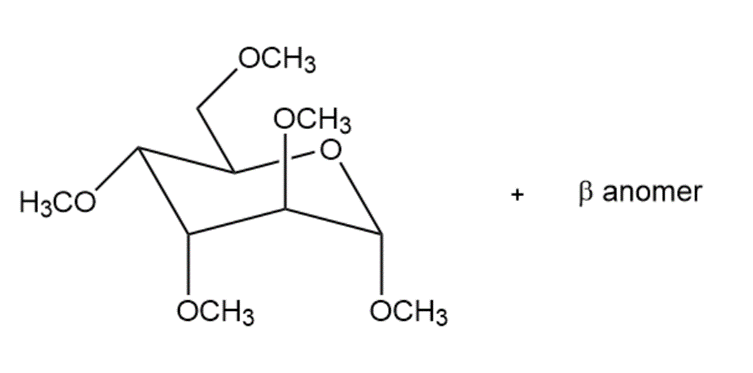
(h)
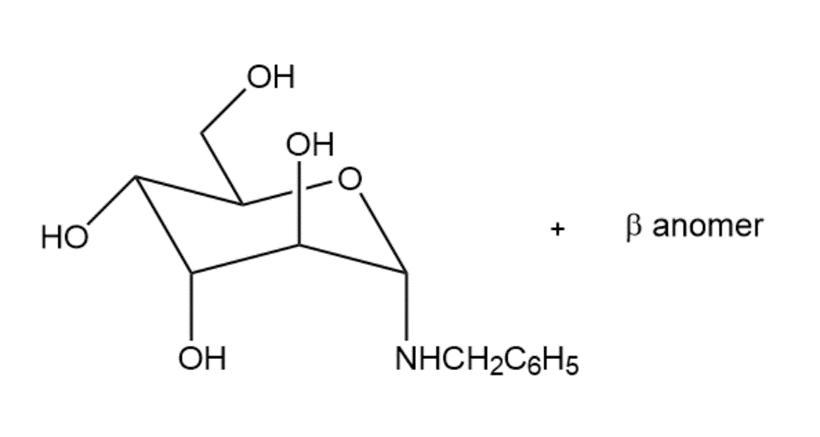
Learning Materials
Features
Discover
Chapter 28: Q50P (page 1148)
Draw the products formed when D-altrose is treated with each reagent.

Answer
(a)

(b)

(c)

(d)

(e)

(f)

(g)

(h)

All the tools & learning materials you need for study success - in one app.
Get started for free
a. Identify the glycosidic linkage in disaccharide C, classify the glycosidic bond as , and use numbers to designate its location.
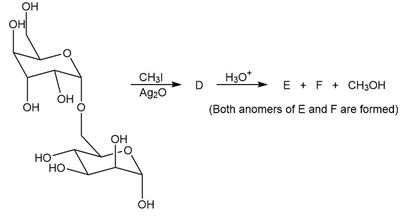
b. Identify the lettered compounds in the following reaction.
What aldoses are formed when the following aldoses are subjected to the Kiliani-Fischer synthesis: (a) D-threose; (b) D-ribose; (c) D-galactose
Draw the structure of a disaccharide formed from two mannose units joined by a glycosidic linkage.
Convert each Haworth projection in Problem 28.14 to a three-dimensional representation using a chair pyranose ring.
Which aldoses are oxidized to optically inactive aldaric acids: (a) D-erythrose; (b) D-lyxose; (c) D-galactose?
What do you think about this solution?
We value your feedback to improve our textbook solutions.