Chapter 28: Question 28.15 (page 1122)
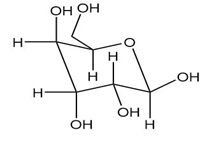
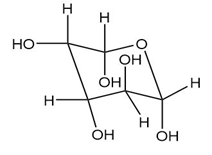
Convert each Haworth projection in Problem 28.14 to a three-dimensional representation using a chair pyranose ring.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 28: Question 28.15 (page 1122)
Convert each Haworth projection in Problem 28.14 to a three-dimensional representation using a chair pyranose ring.
Answer


All the tools & learning materials you need for study success - in one app.
Get started for free
Treating chitin with hydrolyzes its amide linkages, forming a compound called chitosan. What is the structure of chitosan? Chitosan has been used in shampoos, fibers for sutures, and wound dressings.
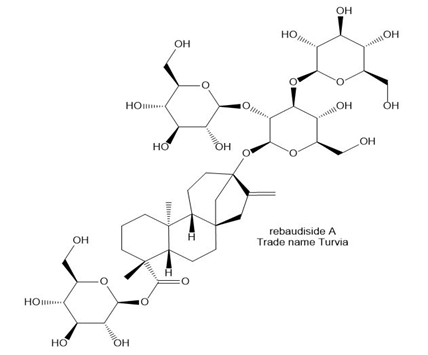
(a)Label all the O atoms that are part of a glycoside in rebaudioside A. Rebaudioside A, marketed under the trade name Truvia, is a sweet glycoside obtained from the stevia plant, which has been used for centuries in Paraguay to sweeten foods. (b)The alcohol or phenol formed from the hydrolysis of glycoside is called an aglycon. What aglycon and monosaccharides are formed by the hydrolysis of rebaudioside A?
As we have seen in Chapter 28, monosaccharides can be drawn in a variety of ways, and in truth, often a mixture of cyclic compounds is present in a solution. Identify each monosaccharide, including its proper D, L designation, draw in a less-than-typical fashion.
(a.)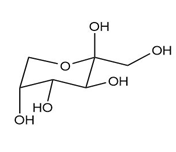
(b.)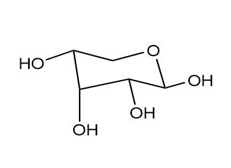
(c.)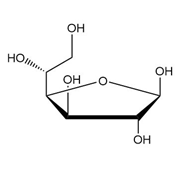
(d.)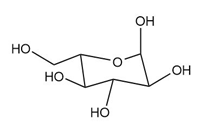
Draw a Fischer projection of the monosaccharide from which each of the following glycosides was prepared.
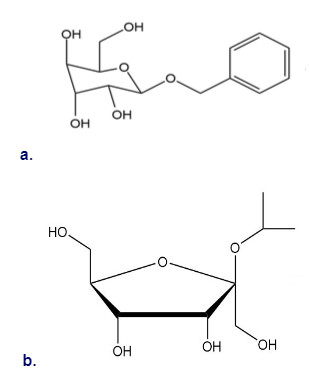
Classify each compound as identical to A or its enantiomer.
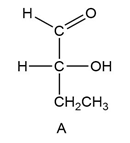
(a.)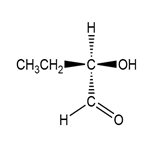
(b.)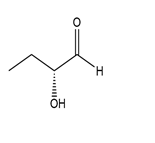
(c.)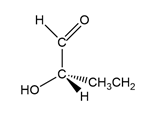
What do you think about this solution?
We value your feedback to improve our textbook solutions.