Chapter 29: Q9P (page 1159)
Draw the products of each reaction.
(a)
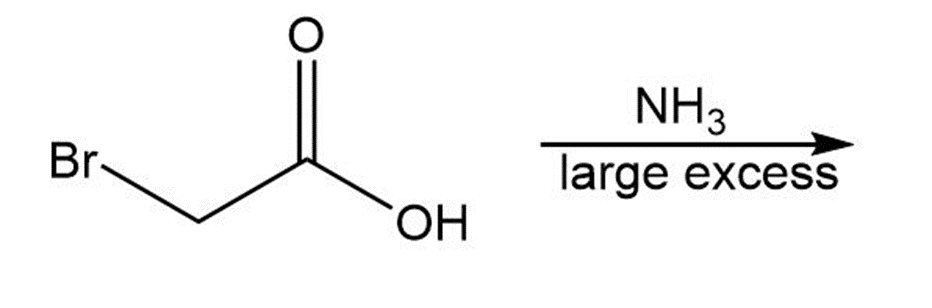
(b)
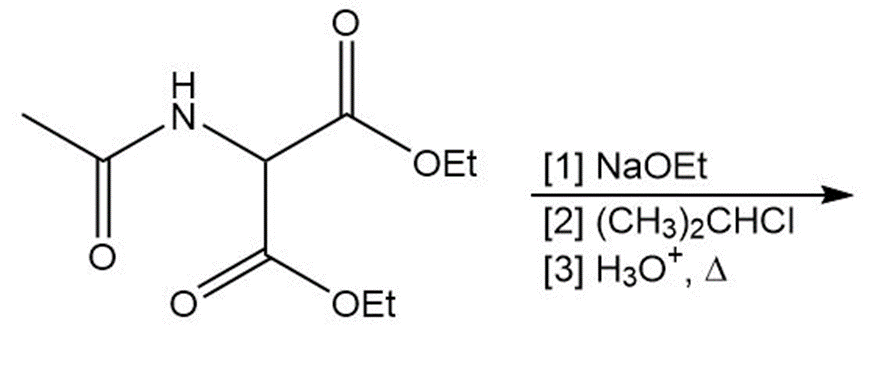
(c)
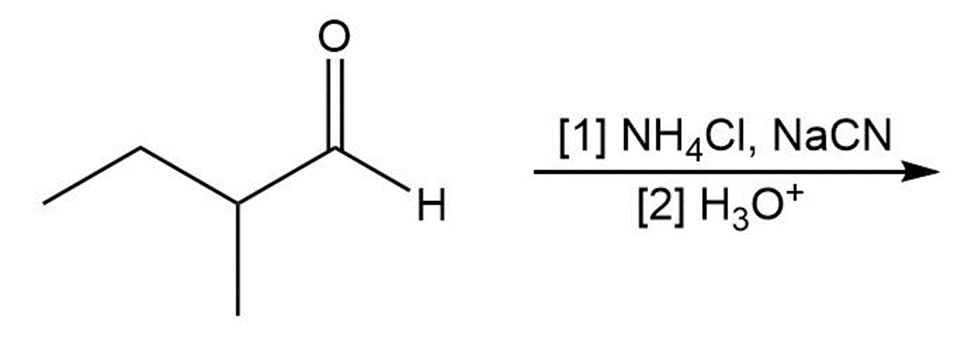
(d)
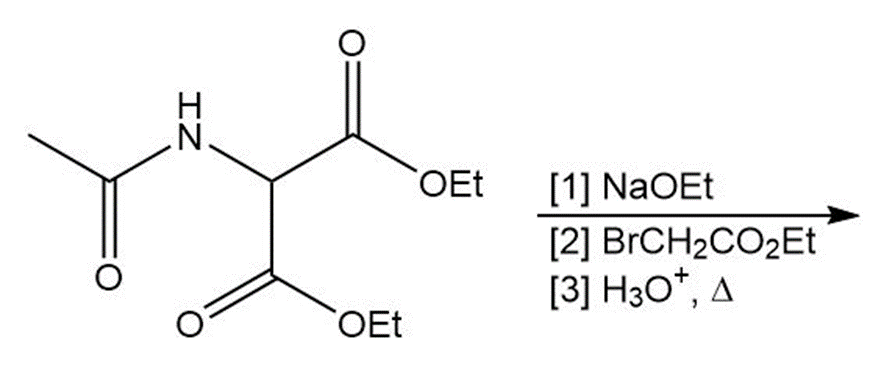
Short Answer
Answer
(a)
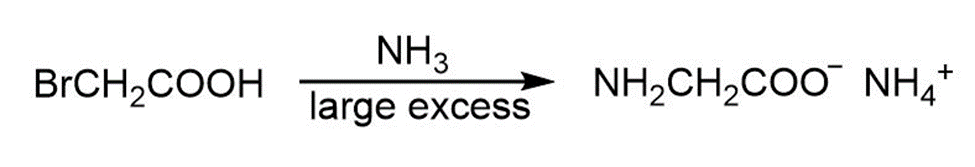
(b)
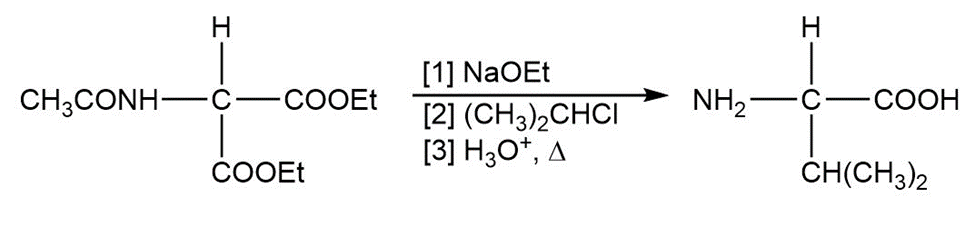
(c)
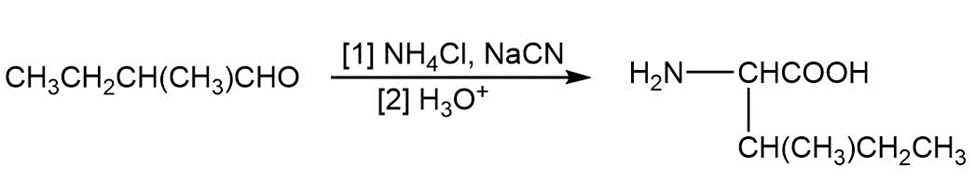
(d)
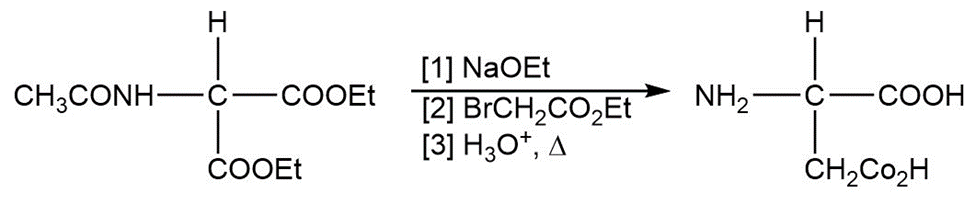
Learning Materials
Features
Discover
Chapter 29: Q9P (page 1159)
Draw the products of each reaction.
(a)

(b)

(c)

(d)

Answer
(a)
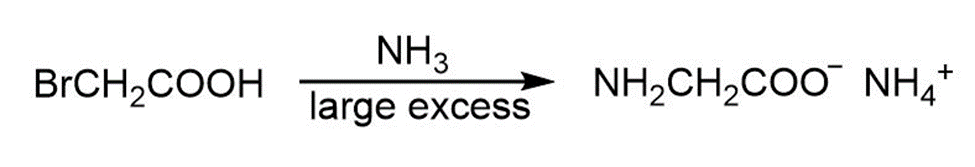
(b)
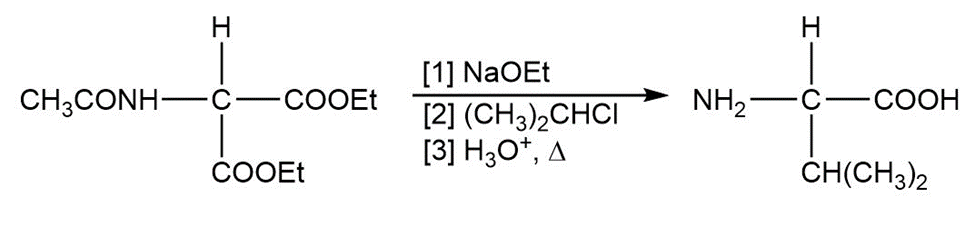
(c)

(d)
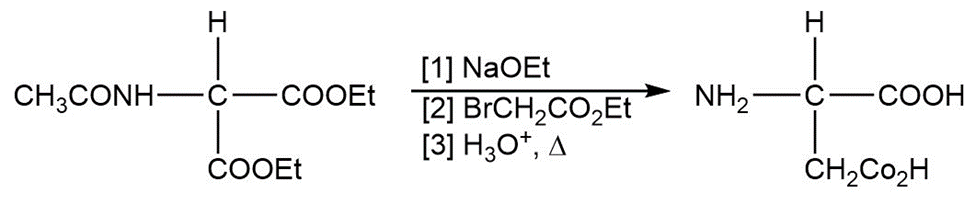
All the tools & learning materials you need for study success - in one app.
Get started for free
What-halo carbonyl compoundis needed to synthesize each amino acid?
(a) Glycine
(b) Isoleucine
(c) Phenylalanine
a. Draw the structure of the tripeptide A-A-A, and label the two ionizable functional groups.
b. What is the predominant form of A-A-A at pH=1?
c. The values for the two ionizable functional groups (3.39 and 8.03) differ considerably from the values of alanine (2.35 and 9.87;see table 29.1). Account for the observed differences.
Draw the amino acids and peptide fragments formed when the decapeptide A-P-F-L-K-W-S-G-R-G is treated with each reagent or enzyme: (a) chymotrypsin; (b) trypsin; (c) carboxypeptidase; (d)
Question: Draw the other three stereoisomers of L-isoleucine, and label the stereogenic centers as R or S.
Devise a synthesis of the following dipeptide from amino acid starting materials.
What do you think about this solution?
We value your feedback to improve our textbook solutions.