Chapter 29: Q1. (page 1155)
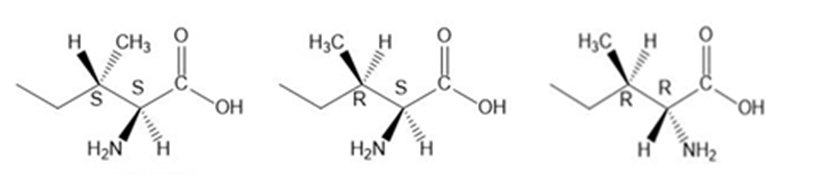
Question: Draw the other three stereoisomers of L-isoleucine, and label the stereogenic centers as R or S.
Short Answer
Learning Materials
Features
Discover
Chapter 29: Q1. (page 1155)
Question: Draw the other three stereoisomers of L-isoleucine, and label the stereogenic centers as R or S.

All the tools & learning materials you need for study success - in one app.
Get started for free
Histidine is classified as a basic amino acid because one of the N atoms in its five-membered ring is readily protonated by acid. Which N atom in histidine is protonated and why?
What form exists at the isoelectric point of each of the following amino acids: (a) valine; (b)leucine; (c)proline; (d) glutamic acid
Draw the organic products formed in each reaction
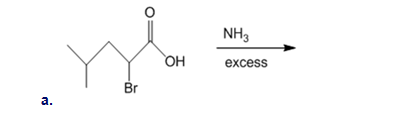
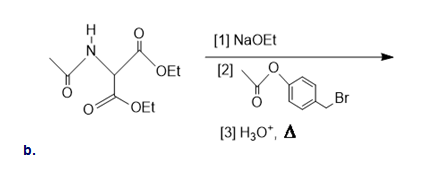
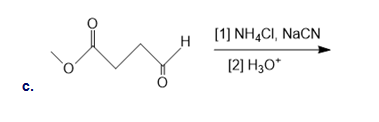
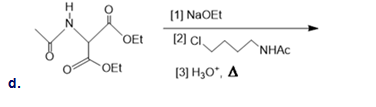
What alkyl halide is needed to synthesize each amino acid from diethyl acetamidomalonate: (a)Asn; (b)His; (c)Trp?
Consider two molecules of a tetrapeptide composed of only alanine residues. Draw the hydrogen bonding interactions that result when these two peptides adopt a parallel β-pleated sheet arrangement. Answer this same question for the antiparallel β-pleated sheet arrangement.
What do you think about this solution?
We value your feedback to improve our textbook solutions.