Chapter 29: 31P (page 1192)
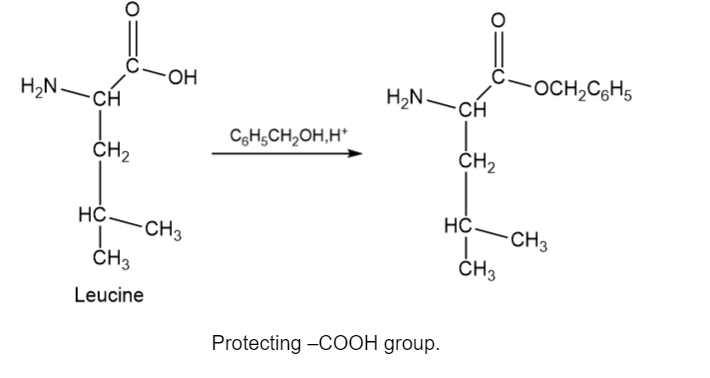
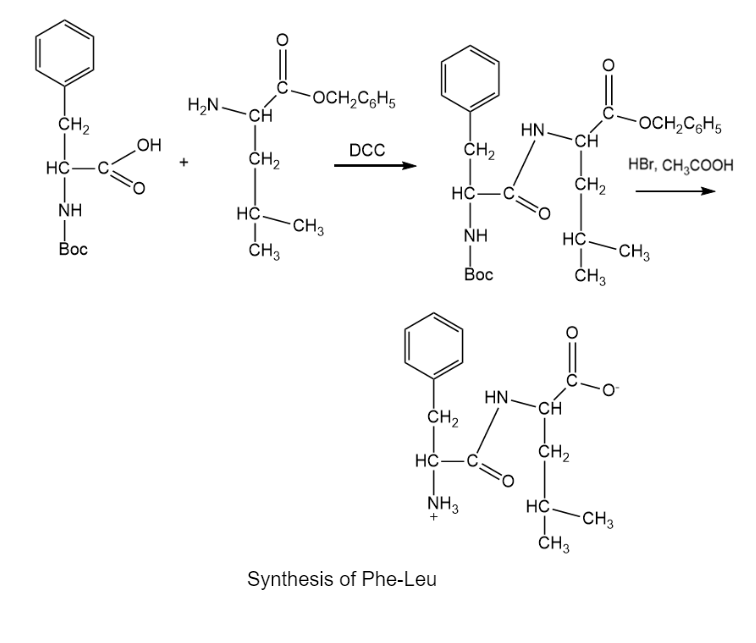
Devise a synthesis of the following dipeptide from amino acid starting materials.
Short Answer
Learning Materials
Features
Discover
Chapter 29: 31P (page 1192)
Devise a synthesis of the following dipeptide from amino acid starting materials.



All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the other three stereoisomers of L-isoleucine, and label the stereogenic centers as R or S.
An octapeptide contains the following amino acids: Arg, Glu, His, Ile, Leu, Phe, Tyr, and Val. Carboxypeptidase treatment of the octapeptide forms Phe and a heptapeptide. Treatment of the octapeptide with chymotrypsin forms two tetrapeptides, A and B. Treatment of A with trypsin yields two dipeptides, C and D. Edman degradation cleaves the following amino acids from each peptide: Glu (octapeptide), Glu (A), Ile (B), Glue (C), and Val (D). Partial hydrolysis of tetrapeptide B forms Ile-Leu in addition to other products. Deduce the structure of the octapeptide and fragments A-D.
(a) What products are formed when each peptide is treated with trypsin? (b) What products are formed when each peptide is treated with chymotrypsin?
[1] Gly–Ala–Phe–Leu–Lys–Ala
[2] Phe–Tyr–Gly–Cys–Arg–Ser
[3] Thr–Pro–Lys–Glu–His–Gly–Phe–Cys–Trp–Val–Val–Phe
Draw the other three stereoisomers of L-isoleucine, and label the stereogenic centers as R or S.
What aldehyde is needed to synthesize each amino acid by the Strecker synthesis?
(a) Valine;
(b) Leucine;
(c) Phenylalanine.
What do you think about this solution?
We value your feedback to improve our textbook solutions.