Chapter 21: 3P (page 817)
Give the IUPAC name for each aldehyde.
a.
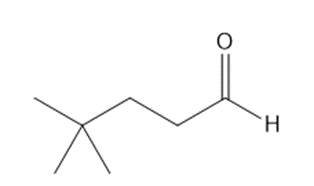
b.
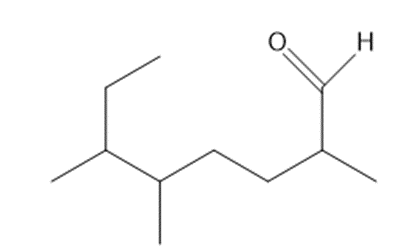
c
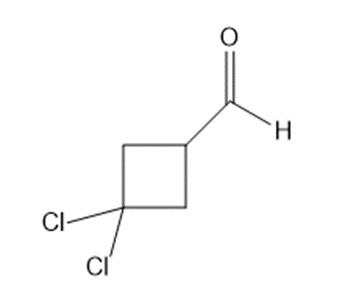
Short Answer
a. 4,4-dimethylpentanal
b. 2,5,6-trimethyloctanal
c. 3,3-dichlorocyclobutanecarbaldehyde
Learning Materials
Features
Discover
Chapter 21: 3P (page 817)
Give the IUPAC name for each aldehyde.
a.

b.

c

a. 4,4-dimethylpentanal
b. 2,5,6-trimethyloctanal
c. 3,3-dichlorocyclobutanecarbaldehyde
All the tools & learning materials you need for study success - in one app.
Get started for free
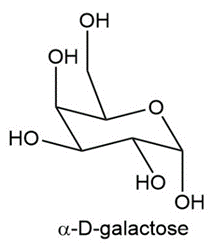
a. How many stereogenic centers are present in -D-galactose?
b. Label the hemiacetal carbon in -D-galactose.
c. Draw the structure of -D-galactose.
d. Draw the structure of the polyhydroxy aldehyde that cyclizes to - and
-D-galactose.
e. From what you learned in Section 21.16B, what product (s) is (are) formed
when -D-galactose is treated with CH3OH and an acid catalyst?
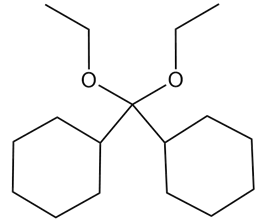
Devise a synthesis of each compound from cyclohexene and organic alcohols. You may use any other required organic or inorganic reagents
a)
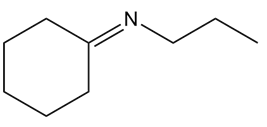
b)
What reagents are needed to convert each compound into butanal?
a.
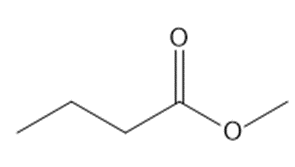
b.
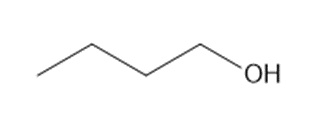
c.
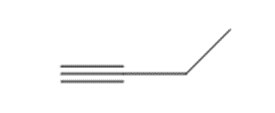
d.
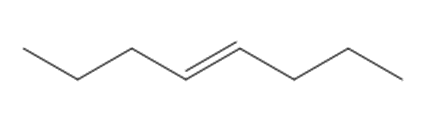
Which compound in each pair forms the higher percentage of gem-diol at equilibrium?
a.
 or
or
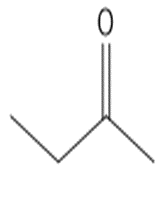
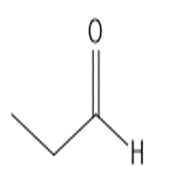
b.
 or
or
Draw the structure of all constitutional isomers that contain a ketone and have molecular formula . Give the IUPAC name for each isomer and state how NMR spectroscopy could be used to distinguish these isomers.
What do you think about this solution?
We value your feedback to improve our textbook solutions.