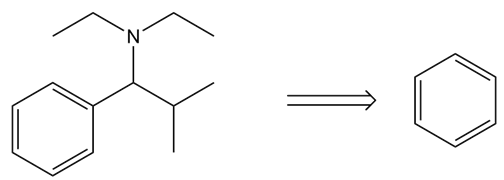
Chapter 21: 19P (page 817)
Draw the products (including stereoisomers) formed when benzaldehyde (C6H5CHO) is treated with each Wittig reagent
a.
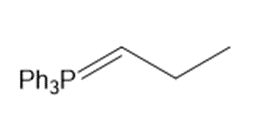
b.
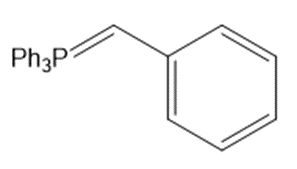
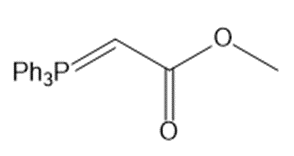
Short Answer
a.
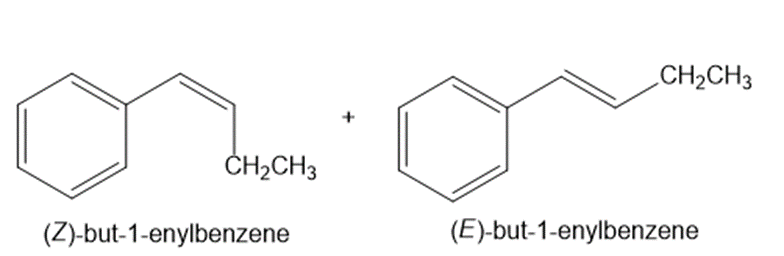
b.
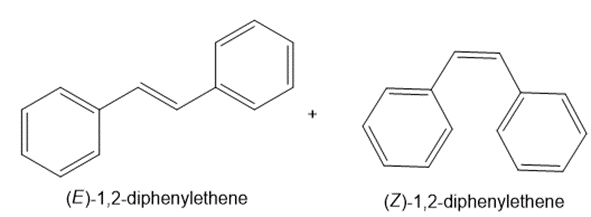
c.
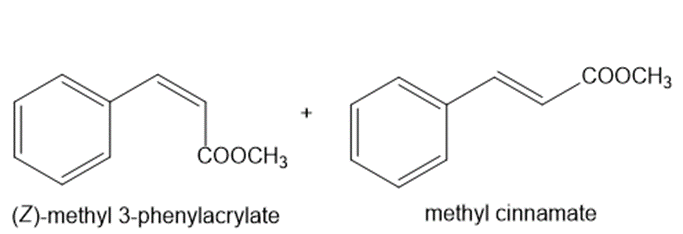
Learning Materials
Features
Discover
Chapter 21: 19P (page 817)
Draw the products (including stereoisomers) formed when benzaldehyde (C6H5CHO) is treated with each Wittig reagent
a.

b.


a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw a stepwise mechanism for the following imine hydrolysis
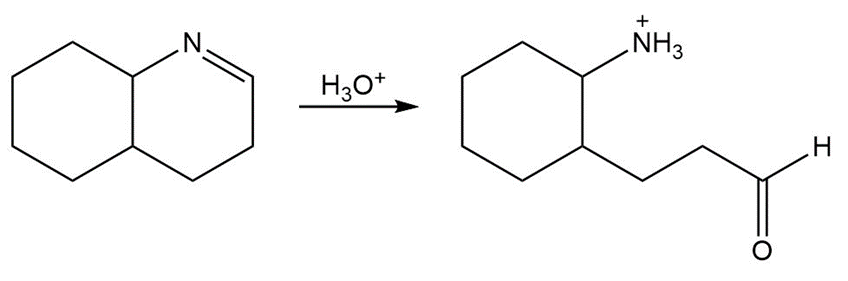
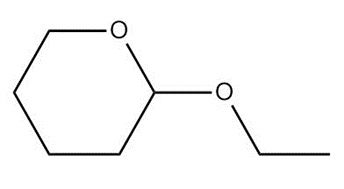
What carbonyl compound and alcohol are formed by hydrolysis of each acetal?
a.
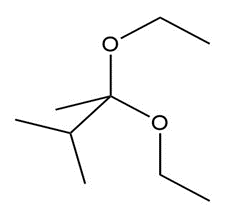
b.
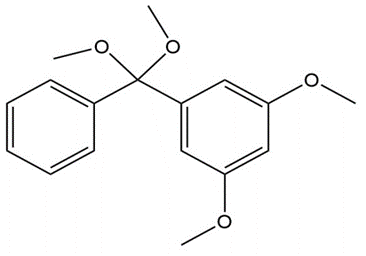
c.
Devise a synthesis of each compound from the given starting materials. You may also use organic alcohols having four or fewer carbons, and any organic or inorganic reagents
a)
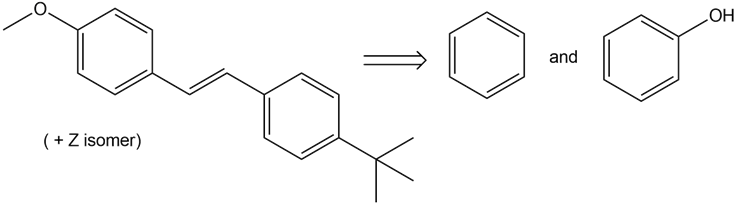
b)
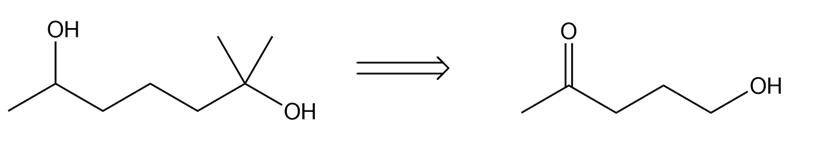
c)
Outline a synthesis of each Wittig reagent from Ph3P and an alkyl halide.
a.
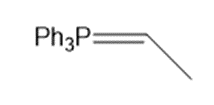
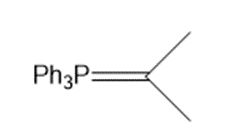
b.
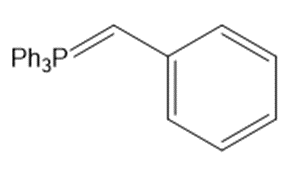
c.
What lactol (cyclic hemiacetal) is formed from intramolecular cyclization of each hydroxy aldehyde?
a.
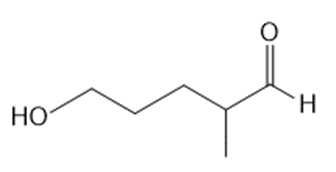
b.

What do you think about this solution?
We value your feedback to improve our textbook solutions.