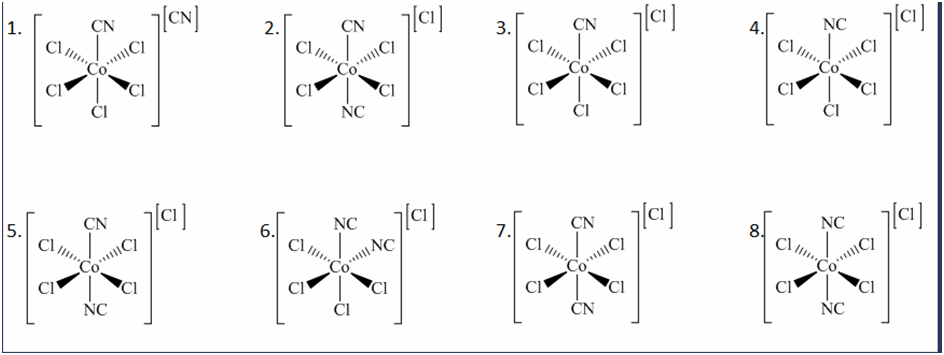
Chapter 19: Q35E (page 1095)
Draw the geometric, linkage, and ionization isomers for \(\left( {CoC{l_5}CN} \right)(CN)\)
Short Answer
Isomers are the chemical species which have the same molecular formula but different arrangement of atoms or ligands in space.