Chapter 19: Q33E (page 1095)
Specify whether the following complexes have isomers.
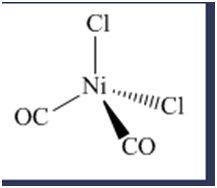
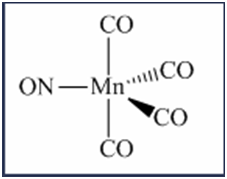
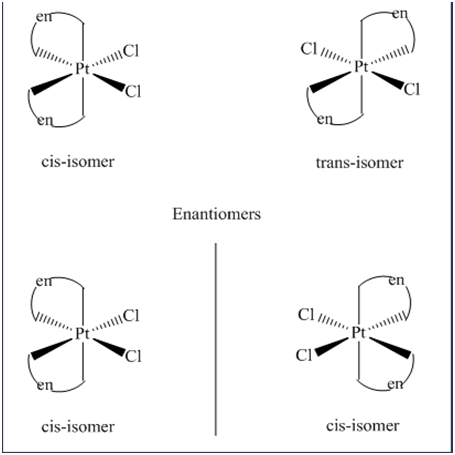
\(\begin{aligned}{}(a)tetrahedral\left( {Ni{{(CO)}_2}{{(Cl)}_2}} \right)\\(b)trigonalbipyramidal\left( {Mn{{(CO)}_4}NO} \right)\\(c)\left( {Pt{{(en)}_2}C{l_2}} \right)C{l_2}\end{aligned}\)
Short Answer
Isomers are the chemical species which have the same molecular formula but different arrangement of atoms or ligands in space. Space isomerism is of two type namely geometrical and optical isomerism.