Chapter 19: Q.27E (page 1094)
Give the coordination numbers and write the formulas for each of the following, including all isomers where appropriate:
(a) tetrahydroxozincate(II) ion (tetrahedral)
(b) hexacyanopalladate(IV) ion
(c) dichloroaurate(I) ion (note that aurum is Latin for “gold”)
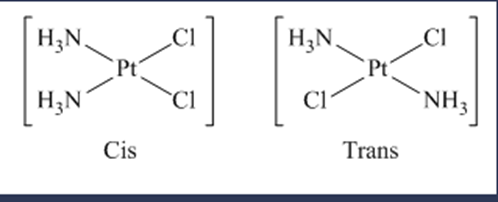
(d) diamminedichloroplatinum(II)
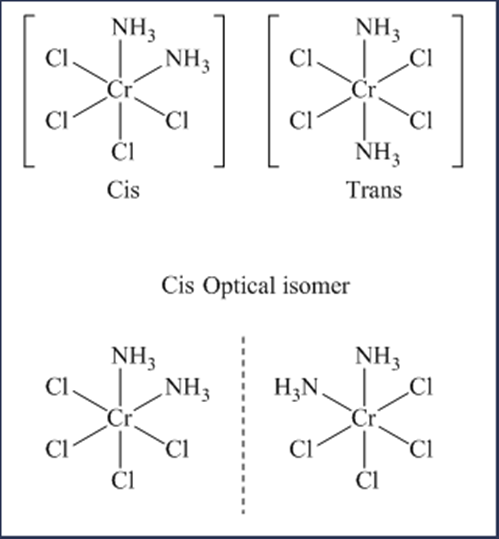
(e) potassium diamminetetrachlorochromate(III)
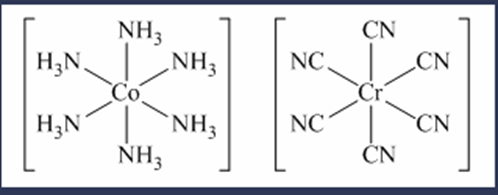
(f) hexaamminecobalt(III) hexacyanochromate(III)
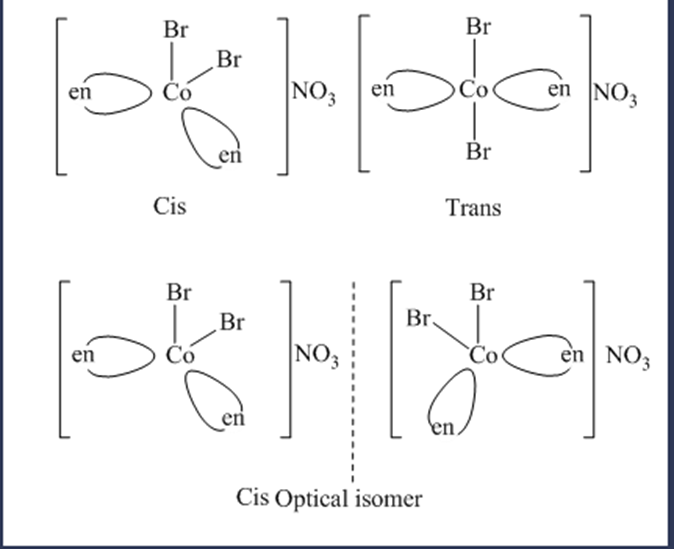
(g) dibromobis(ethylenediamine) cobalt(III) nitrate
Short Answer
The number of ligands attached to the central metal ion attached through the secondary valencies and are present inside the coordination sphere is called coordination number of central metal ion.