Chapter 4: Q74E (page 223)
How many molecules of C2H4Cl2 can be prepared from 15 C2H4molecules and 8 Cl2molecules?
Short Answer
8 molecules of\({C_2}{H_4}C{l_2}\)can be prepared from 15 C2H4molecules and 8 Cl2molecules
Learning Materials
Features
Discover
Chapter 4: Q74E (page 223)
How many molecules of C2H4Cl2 can be prepared from 15 C2H4molecules and 8 Cl2molecules?
8 molecules of\({C_2}{H_4}C{l_2}\)can be prepared from 15 C2H4molecules and 8 Cl2molecules
All the tools & learning materials you need for study success - in one app.
Get started for free
A sample of gallium bromide, GaBr2, weighing 0.165 g was dissolved in water and treated with silver nitrate, AgNO3, resulting in the precipitation of 0.299 g AgBr. Use these data to compute the %Ga (by mass) GaBr2.
What is the percent yield of a reaction that produces 12.5g of the gas freon CF2Cl2 from 32.9 g of CCl4 and excess HF.
\(CC{l_4} + 2HF \to C{F_2}\)
Outline the steps needed to determine the limiting reactant when 0.50 mol of Cr and 0.75 mol of H3PO4 react according to the following chemical equation.\(2Cr + 2{H_3}P{O_4} \to 2CrP{O_4} + 3{H_2}\). Determine the limiting reactant.
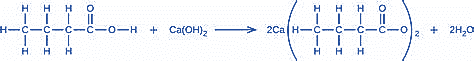
What mass of Ca(OH)2 will react with 25.0 g of propionic acid to form the preservative calcium propionate according to the equation?
Balance the following equations:
\(\begin{array}{l}\left( a \right)\,Ag\left( s \right) + {H_2}S\left( g \right) + {O_2}\left( g \right) \to A{g_2}S\left( s \right) + {H_2}O\left( l \right)\\\left( b \right)\,{P_4}\left( s \right) + {O_2}\left( g \right) \to {P_4}{O_{10}}\left( s \right)\\\left( c \right)\,Pb\left( s \right) + {H_2}O\left( l \right) + {O_2}\left( g \right) \to Pb{\left( {OH} \right)_2}\left( s \right)\\\left( d \right)\,Fe\left( s \right) + {H_2}O\left( l \right) \to F{e_3}{O_4}\left( s \right) + {H_2}\left( g \right)\\\left( e \right)\,S{c_2}{O_3}\left( s \right) + S{O_3}\left( g \right) \to S{c_2}{\left( {S{O_4}} \right)_3}\left( s \right)\\\left( f \right)\,C{a_3}{\left( {P{O_4}} \right)_2}\left( {aq} \right) + {H_3}P{O_4}\left( {aq} \right) \to Ca{\left( {{H_2}P{O_4}} \right)_2}\left( {aq} \right)\\\left( g \right)\,Al\left( s \right) + {H_2}S{O_4}\left( {aq} \right) \to A{l_2}{\left( {S{O_4}} \right)_3}\left( s \right) + {H_2}\left( g \right)\\\left( h \right)\,TiC{l_4}\left( s \right) + {H_2}O\left( g \right) \to Ti{O_2}\left( s \right) + HCl\left( g \right)\end{array}\)
What do you think about this solution?
We value your feedback to improve our textbook solutions.