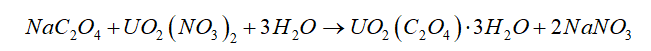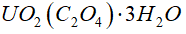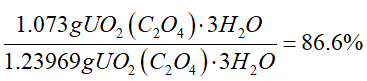Chapter 4: Q73E (page 226)
Uranium can be isolated from its ores by dissolving it as UO2(NO3)2, then separating it as solid UO2(C2O4). Addition of 0.4031 g of sodium oxalate, NaC2O4, to a solution containing 1.481 g of uranyl nitrate, UO2(NO3)2, yields 1.073 g of solid
\(Na{C_2}{O_4} + U{O_2}{\left( {N{O_3}} \right)_2} + 3{H_2}O \to U{O_2}\left( {{C_2}{O_4}} \right) \cdot 3{H_2}O + 2NaN{O_3}\)
Short Answer
NaC2O4 is the limiting reactant.
The percent yield is 86.6 %.