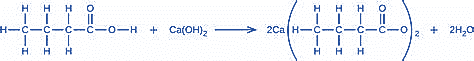
Chapter 4: 4.3 CYL (page 183)
Which solution could be used to precipitate barium ion Ba2+, in a water sample, Sodium chloride, sodium hydroxide, or sodium sulfate. What is the formula for the expected precipitate?
Short Answer
Sodium Sulfate is used to precipitate a barium ion.
The formula for the expected precipitate is BaSO4.