Chapter 20: Q49E (page 1145)
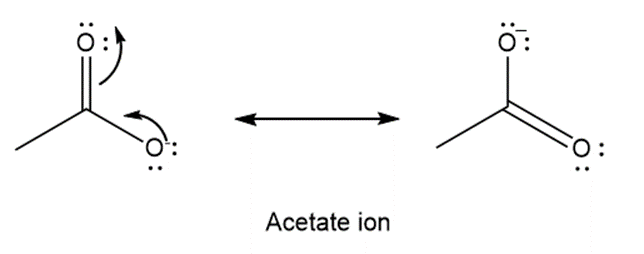
Write the two-resonance structures for the acetate ion.
Short Answer
Resonance structures of acetate ion are as shown in the figure:
Learning Materials
Features
Discover
Chapter 20: Q49E (page 1145)
Write the two-resonance structures for the acetate ion.
Resonance structures of acetate ion are as shown in the figure:

All the tools & learning materials you need for study success - in one app.
Get started for free
Write two complete, balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures.
Ethylammonium chloride is added to a solution of sodium hydroxide.
Identify any carbon atoms that change hybridization and the change in hybridization during the reactions in Exercise 20.39.
Write the structures for all the isomers of the \(--{C_5}{H_{11}}\) alkyl group.
Explain why these two molecules are not isomers:
Explain why it is not possible to prepare a ketone that contains only two carbon atoms.
What do you think about this solution?
We value your feedback to improve our textbook solutions.