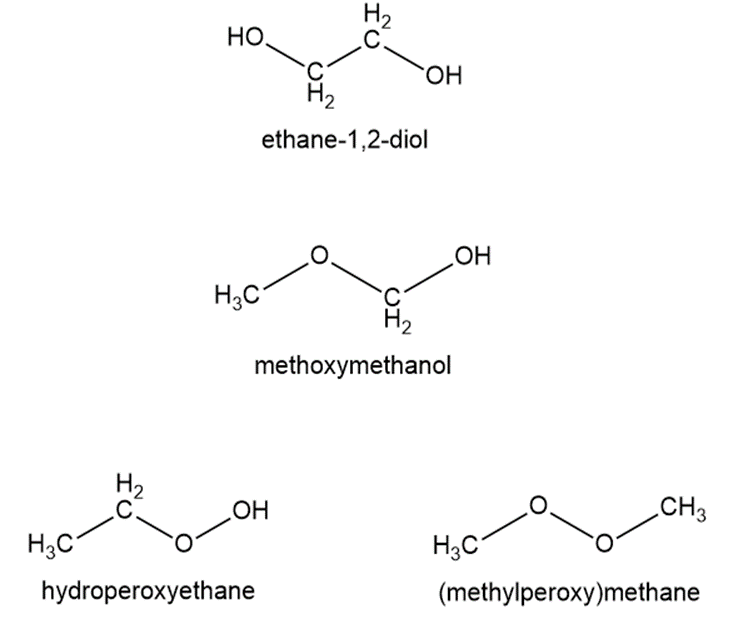
Chapter 20: Q35 E (page 1143)
Write the condensed structures of all isomers with the formulaC2H6O2. Label the functional group (or groups) of each isomer.
Short Answer
Learning Materials
Features
Discover
Chapter 20: Q35 E (page 1143)
Write the condensed structures of all isomers with the formulaC2H6O2. Label the functional group (or groups) of each isomer.

All the tools & learning materials you need for study success - in one app.
Get started for free
Write the condensed structures of both isomers with the formula C2H6O. Label the functional group of each isomer.
Name the following molecule:
Write structures for the three isomers of the aromatic hydrocarbon xylene,\({C_6}{H_4}{\left( {C{H_3}} \right)_2}\)
Explain why these two molecules are not isomers:
Write Lewis structures and IUPAC names for all isomers of \({C_4}{H_9}Cl.\)
What do you think about this solution?
We value your feedback to improve our textbook solutions.