Chapter 20: Q34 E (page 1143)
Write the condensed structures of both isomers with the formula C2H6O. Label the functional group of each isomer.
Short Answer
Isomers of the given compound:
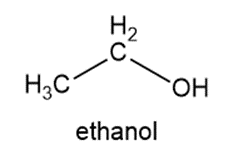
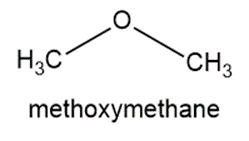
Learning Materials
Features
Discover
Chapter 20: Q34 E (page 1143)
Write the condensed structures of both isomers with the formula C2H6O. Label the functional group of each isomer.
Isomers of the given compound:


All the tools & learning materials you need for study success - in one app.
Get started for free
Teflon is prepared by the polymerization of tetrafluoroethylene. Write the equation that describes the polymerization using Lewis symbols.
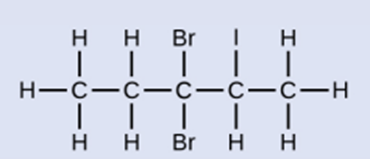
Name the following molecule:
How does the hybridization of the substituted carbon atom change when alcohol is converted into an aldehyde? An aldehyde to a carboxylic acid?
Write two complete, balanced equations for each of the following reactions, one using condensed formulas and one using Lewis structures.
Ethylammonium chloride is added to a solution of sodium hydroxide.
Alcohols\(A,B\) , and \(C\) all have the composition \({C_4}{H_{10}}O.\) Molecules of alcohol A contain a branched carbon chain and can be oxidized to an aldehyde; molecules of alcohol B contain a linear carbon chain and can be oxidized to a ketone; and molecules of alcohol C can be oxidized to neither an aldehyde nor a ketone. Write the Lewis structures of these molecules.
What do you think about this solution?
We value your feedback to improve our textbook solutions.